Abstract
It is known that myocardial infarction (MI) causes damages to the heart tissue and that present medical therapies, such as medication, stenting and coronary artery bypass surgery, cannot recover the injured heart. Fortunately, advances in stem cell research have brought hope of full heart recovery for myocardial ischemia patients. There have been many studies using cell therapies for myocardial ischemia, from preclinical trials to clinical trials. However, the biggest concern is the effect of transplanted cells in myocardial recovery. This review will focus on analyzing both the positive and negative effects of transplanted cells in myocardial recovery to better understand the underlying biological mechanisms and ways to evaluate safety and efficacy of cell transplantation in myocardial ischemia treatment.
Introduction
Myocardial infarction (MI) kills cardiac cells and forms scars in the heart tissue.
Although the formation of scars helps the injured heart cope with damages quickly, protects healthy tissue from further damage and prevents a cascade of adverse uncontrollable events Azouz et al., 2004, the biochemical reactions during scar formation remain unclear. In fact, the physical and functional properties during heart tissue scarring is similar to those of normal tissues.
Scarring has negative effects on the structure and activity of the infarcted heart Cregg et al., 2014Silver and Miller, 2004Xu et al., 2004.
Current treatments such as lifestyle changes, medication, stent intervention or artery bypass surgery can only support the heart to slow the failure process, but cannot recover damaged heart tissues. The successful rate of all heart transplantation was very low. The number of novel postnatal heart tisue was low too and it was around 1% of total myocardium and decreased with age Garbern and Lee, 2013.
Nowadays with the development of regenerative medicine, cell therapy can be expected to completely restore the structure and function of the damaged heart. Cell transplantations into ischemic areas have been investigated in small animal models Avolio et al., 2015Kim et al., 2015Tang et al., 2015, large animal models Kanazawa et al., 2015Yee et al., 2014 and in clinical trials Karantalis et al., 2012Perin et al., 2015. Various cell types have been used in implantation, including mesenchymal stem cells (MSCs) Kocher et al., 2001, induced pluripotent stem cells (iPSCs) Cantz and Martin, 2010, cardiac progenitor cells (CPCs) Garbern and Lee, 2013, cells derived from fetal tissue and adult cardiomyocytes Soonpaa et al., 1994Zhang et al., 2001, skeletal myoblasts Menasche et al., 2001, muscle cells Condorelli et al., 2001, embryonic-derived endothelial cells (ECs) Condorelli et al., 2001, bone marrow-derived immature cells Hattan et al., 2005, fibroblasts Galli et al., 2005, smooth muscle cells (SMCs) Harada et al., 2016 and bone marrow-c-kit positive and negative progenitor cells Fazel et al., 2008Fernandez-Aviles et al., 2004. In spite of the fact that results with the above have varied, they were largely similar in that there is a positive impact of cell transplantation on the recipients. It is important to understand how the transplanted cells act in heart wound healing and their efficacy. This review will focus on those issues.
Effects of transplanted cells
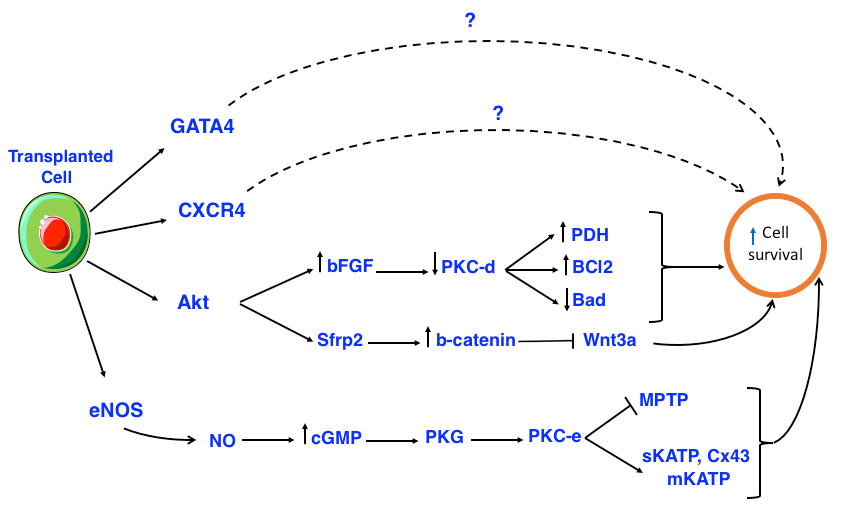
Most likely transplanted cells reduce negative remodeling by reducing the stiffness of the ventricular wall scar and restoring the lost heart muscle. Proposed mechanisms for this process include: (1) the transplanted cells secrete paracrine factors to protect the cells from apoptosis, mobilize the available cardiac stem cells, activate their proliferation and differentiation into heart cells, partake in neovascularization, reduce scar formation and limit inflammation; (2) the transplanted cells can fuse with host graft; (3) the transplanted cells can differentiate into cardiac muscle cells ( Figure 1 ).
Secretion of growth factors
Secretion of implanted cells plays an important role in repairing heart tissue damage. Adult stem cells, particularly MSCs, after transplantation can release a variety of cytokines, chemokines and growth factors involved in heart repair process Deb et al., 2008Li et al., 2012Loffredo et al., 2011. These factors, in turn, induce neighboring stem cells to secrete cytokines and induce changes in the microenvironment which promote proliferation and differentiation of stem cells Behfar et al., 2002Doyle et al., 2016Gude et al., 2015Kinnaird et al., 2004. In particular, properties such as myocardial protection and neovascularization of paracrine factors currently have been most widely studied. Moreover, the secreted factors also impact positively on the inflammation process, fibrogenic process, heart metabolism, heart contractions and/or endogenous cardiac regeneration. These effects may occur in different ways and are dependent on the microenvironment after infarction. These factors may also act in an autocrine fashion, impacting the cells which secrete them Deb et al., 2008.
Protection of heart tissue
The immediate impact of stem cells after grafting into the heart muscle is to release cytoprotective molecules to increase myocardial viability. These molecules inhibit apoptosis by activating AKT/PKB signaling pathway (Rosenberg et al., 2012; Yang et al., 2012). Some studies have shown that Aktoverexpressing MSCs and exosomes secreted from CXCR4-overexpressing or GATA4-overexpressing MSCs are able to significantly prevent apoptosis, thereby reducing infarction size Kang et al., 2015Noiseux et al., 2006Yu et al., 2015. Other studies have shown that elements secreted from grafted bone marrow stem cells induce cardiomyocyte protection in the ischemic region Broughton and Sussman, 2016Dai et al., 2008Xu et al., 2007. Cardiomyocytes differentiated from bone marrow monocytes (BM-MNCs) cultured under hypoxic conditions show they have inhibitory effects on apoptosis and can reduce infarction size when transplanted into the body Kubal et al., 2006. When heart muscle cells and BM-MNCs obtained from the same patients were co-cultured, cell necrosis and apoptosis were significantly reduced; cell protection, however, did not occur when heart muscle cells with co-cultured with ECs or keratinocytes Yoon et al., 2005.
Moreover, transplanted Akt-expressing MSCs also expressed secreted frizzled related protein 2 (Sfrp2), which increases cellular total -catenin of cardiomyocytes. The -catenin protected cardiomyocytes of newborn rats were stable against hypoxia and reoxygenation-induced apoptosis by blocking the pre-apoptotic effects of Wnt3a Mirotsou et al., 2007Zhang et al., 2009. AktMSCs regulated the increase of vascular endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF), hepatocyte growth factor (HGF), insulin-like growth factor-1 (IGF-1), and thymosin 4 (TB4). In turn, these factors promoted neovascularization, protection of myocardium and cardiac contractility Gnecchi et al., 2006 ( Figure 2 ).
The overexpression of bFGF can enhance the recovery of contractile function and reduce infarction area after reperfusion. In fact, bFGF reduces the protein kinase C delta (PKC-d) displacements but does not affect PKC-alpha (PKC-a), PKC-epsilon (PKC-e), or PKC-zeta (PKC-z). PKC-d reduction protects heart cells and decreases the number of dead cells. In addition, bFGF has been shown to be related to the MAPK/ERK signaling pathway in heart cell protection, although the mechanism remains unclear Baines et al., 2002House et al., 2007Padua et al., 1998Rose et al., 2010Srisakuldee et al., 2014. Akt activates a number of substrates, including members of the B-cell lymphoma 2 (Bcl-2) protein family, glycogen synthase kinase 3 beta (GSK-3) and endothelial nitric oxide synthase (eNOS). Nitric oxide (NO) synthesized from eNOS activates PKG through intracellular cGMP increasing. The substrates for protein kinase G (PKG) are thought to include SR regulation proteins and phospholamban, which promotes SR Ca2+ absorption thereby reducing the overload of cytosolic Ca2+. PKG is the final component of the signal transduction leading to activation of PKC-epsilon (PKC-e) mitochondrial pool. Activated PKC-e in turn activates mitochondrial
ATP-dependent potassium channels (mKATP), promoting the reactive oxygen species (ROS) formation ( Figure 2 ).
The inhibition of mitochondrial permeability transition pore (MPTP) may occur as a result of PKC-e activation. Sarcolemmal KATP (sKATP) and mitochondrial connexin-43 (Cx43) are also considered components of the pre-regulation mechanism. The formation of ROS and reactive nitrogen species (RNS) are the results of mKATP opening and are mandatory components of the signaling cascade. It seems that ROS /RNS signaling is related to activation of kinases, such as p38, MAPK, PKC and JAK/STAT Ferdinandy et al., 2007.
The heart protects extracellular ligands, including adenosine, bradykinin and opioids, while activating multiple kinases, including p42/p44, MAPK/ERK, PI3K/Akt and PKC. The substrates of PKC related to the regulating protective mechanisms are still unknown. There is also evidence that the activation of PKG might occur as part of the post-regulating mechanism downstream of Akt/NO/cGMP, and may play a role in promoting the absorption of sarcoplasmic reticulum Ca2+. It is known that activation of Akt inhibits GSK-3 and members of the chemokine (C-C motif) ligand 2 (Ccl-2) protein family, leading to inhibition of the formation of mKATP-mediated MPTP Ferdinandy et al., 2007.
Normally, PKC exists in a non-activated state until it is stimulated by phospholipid diacyl glycerol (DAG)-derived second messengers. Intracellular ROS translocates to the subcellular target locations, including mitochondria, sarcolemmal membrane and gap junctions. Indeed, sKATP-mediated phosphorylation leads to shortening of the action potential, reducing the Ca2+ overload during ischemia. Moreover, PKC-mediated phosphorylation of Cx43 reduces connexon permeability and prevents the expansion of damages between co-joined cells. PKC-mediated phosphorylation opens the mKATP, conserves mitochondria function and forms local ROS. In turn, ROS may initiate PKC signaling via positive feedback. MPTP inhibition can occur by both direct mechanisms (e.g. via PKC phosphorylation) and indirect mechanisms (e.g. via mKATP opening which reduces cell death after infarction). Receptor for activated c-kinase (RACK) treatment and erythropoietin also promote PKC activation, resulting in heart cell protection Bearzi et al., 2007Budas et al., 2007.
In summary, during ischemia reperfusion activated PKC-e in turn activates sKATP and mKATP, phosphorylates Cx43 and inhibits MPTP, leading to cell protection. However, activated PKC-d inhibits pyruvate dehydrogenase (PDH), decreases Bcl-2, increases Bcl-2-associated death promoter (Bad) protein, causing apoptosis and necrosis. Thus, transplanted cells protect myocardium by secreting various factors that activate PKC-e and inhibit PKC-d.
Mobilization of resident stem cells
The discovery and recognition of the existence of cardiac stem cells led to a shift by researchers and physicians to explore the use of stem cells for new cardiovascular disease therapies Bearzi et al., 2009Hosoda et al., 2009Urbanek et al., 2006. In the heart, cardiac stem cells (CSCs) have existed in their niches. Normally, these cells have kept "silent" in their niches, and have been nourished and controlled by feeding cells within the niches. Upon receiving trigger signals, CSCs undergo a symmetrical or asymmetrical proliferation; they separate from their niches and migrate to areas where they were needed to replace damaged or dying heart cells. However, the number of CSCs are very rare, at approximately 1 stem cell per 30,000 heart cells Beltrami et al., 2003Urbanek et al., 2006. Therefore, based on the normal growth rate of resident CSCs, when infarction occurs the number of cells needed to be replaced is much greater than the number of available cells, leading to a lack of intrinsic cardiac stem cells for replacement. This means that while the injured heart may be delayed in damages in the short term, heart failure cannot be reversed in the long term Urbanek et al., 2003.
Studies have shown that when MSCs are grafted into the body in the absence of oxygen, they release HGF and IGF-1 to mobilize and activate resident CSCs Gómez-Mauricio et al., 2016Linke et al., 2005. Besides MSCs, endothelial progenitor cells (EPCs) play an important role. EPCs activate cardiac regenerative pools and promote the migration, proliferation and differentiation of CSCs (via secretion of cytokines such as VEGF, IGF-1 and SDF-1) Urbich et al., 2005. These factors induce interstitial CSCs to move through the myocardium to necrotic myocardium and scar areas. There, they divide and differentiate into heart cells and become involved in the process of new blood vessel formation Bian et al., 2014Hosoda et al., 2009Tillmanns et al., 2008. CSC invasion to scar tissue is believed to be related to matrix metalloproteinase (MMP)-9 and -14 mediated regulation Bax et al., 2012Huang et al., 2011Rota et al., 2008.
Neovascularization
MSC transplantation improves reperfusion efficiency by increasing the formation of new blood vessels. However, MSCs are rarely present in the new vessels; their main activity is to secrete angiogenic factors, such as VEGF, bFGF, angiopoietin-1, NO and HGF Kinnaird et al., 2004Zhao et al., 2010. These factors increase the permeability of the capillary wall, activate MMP, promote the proliferation and migration of ECs and vascular smooth muscle cells (VSMCs), and form new vessels in the lesions Jiang et al., 2006Louis and Zahradka, 2010Nagaya et al., 2004. Besides MSCs, transplanted EPCs also enhance new vessel formation by releasing VEGF and stromal cell-derived factor 1 (SDF-1) into
the cellular matrix, thereby promoting the migration and maturation of EPCs into ECs Cochain et al., 2013Urbich et al., 2005Wu et al., 2006. Angiogenic cells have been implanted as hydrogel supplying scaffolds to increase microvasculature along infarction areas, thereby significantly improving coronary blood flow and ejection fraction after MI Kim et al., 2012Leblanc et al., 2013Levit et al., 2013. In addition to angiogenesis factors, adult stem cells secrete TB4 and erythropoietin (EPO) Lv et al., 2015Smart et al., 2007. TB4 induces
the proliferation and circuit network formation of epicardium-derived cells (EPDCs) and is involved in the intermediate PKC signaling pathway Smart et al., 2013Smart et al., 2007. Meanwhile, granulocyte colony stimulating factor (GCSF) and EPO mobilize hematopoietic stem cells (HSCs) and EPCs from bone marrow for angiogenesis by activating Janus-activated tyrosine kinase 2 (JAK2) through STAT, PI3K/Akt and MAPK signaling pathways (Nagai and Komuro, 2012).
Impact on extracellular matrix (ECM) and reduction of scar formation
After infarction, scars are formed to replace injury tissues damaged by myocardial ischemia. During ischemia, the ECM secretion process is disordered due to heart cells dying and the body's self-regulation. These impact the thickness of the developing scar, leading to an effect on the contraction of the surrounding heart tissue. A decrease in the ECM makes the ventricular wall thinner, causing left ventricular (LV) rupture while an increase in the ECM enhances fibrosis, leading to heart failure over time Zamilpa and Lindsey, 2010.
From studies, it has been observed that grafted cells are capable of regulating scar formation through inhibition of fibroblast proliferation; furthermore, it has been shown that paracrine factor secretion alters ECM to improve cardiac functions Berry et al., 2006. Transplanted MSCs reduce myofibroblasts through releasing MMPs Almalki and Agrawal, 2016Mias et al., 2009. In MI rat models, implanted MSCs reduce the expression of collagen types I and III, tissue inhibitor of metalloproteinase-1 (TIMP-1), MMP2, MMP9, and transforming growth factor-beta (TGF-beta) Nagaya et al., 2005Xu et al., 2005. It is interesting that transplanted MSC cardiomyocyte (MSC-CM) also express the ability to reduce the scarring process by downregulating fibroblast proliferation and inhibiting the expression of collagen type I and type III in myofibroblasts Ohnishi et al., 2007aOhnishi et al., 2007b. In addition, embryonic stem cell cardiomyocyte (ESC-CM) also show the ability to reduce scarring after MI Leor et al., 2007. In sheep, MSCs injected one hour after MI also show changes in MMP-1, -2, -3, -7, -9, -13, membrane type 1-MMP (MT1-MMP), and TIMPs-1, -2, -4, at the border zone and infarct zone Dixon et al., 2009.
Limiting inflammation
After MI, the inflammatory process is needed to mobilize immune cells to clear out dead heart cells and debris, and to stimulate ventricular remodeling Frangogiannis, 2012Frangogiannis et al., 2002. However, prolonged inflammatory responses would be detrimental to the remodeling process and ventricular function due to heart cell loss leading to negative impacts on ECM as well as formation of new vessels Frangogiannis et al., 2002. Transplanted cells, such as MSCs or MSC-CMs, are able to limit the inflammatory process in the injured tissue. They weaken the proliferation of inflammatory CD68+ cells, decrease the expression of monocyte chemoattractant protein (MCP-1), increase the expression of genes involved in DNA repair, increase antioxidant enzymes and stimulate detoxifier systems, thereby improving cardiac function Fuse et al., 2001Ohnishi et al., 2007bRamalho-Santos et al., 2002.
Transplanted MSCs increase the number of M2 macrophages which were at the anti-inflammatory stage. The mechanism of this process is related to a variety of paracrine factors derived from transplanted MSCs, such as CCL2, galectin-1, interferon-γ, interleukin (IL)-1β, indoleamine-2,3-dioxygenase, IL-4, IL-6, IL-10, IL-13, prostaglandin E2 (PGE2), tumor necrosis factor (TNF)-α, nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB), NO, heme oxygenase-1, HGF, and TGF-beta Ben-Mordechai et al., 2013Bernardo and Fibbe, 2013Du et al., 2008. Normally, the pro-inflammatory cytokines (e.g. NF-κB, IL-6 and TNF-) poison the heart muscle cells, causing detrimental effects on cardiac contractile function. Transplanted MSCs have been shown to inhibit activation of NF-κB, reduce production of TNF- α and IL-6, and increase the expression of anti-inflammatory cytokine IL-10, thereby limiting inflammation after infarction Du et al., 2008Onai et al., 2007. Moreover, the increase of indoleaminepyrrole 2,3-dioxygenase (IDO) and PGE2 secretion also reduces T-cell activity and NK cell proliferation Glennie et al., 2005Nauta et al., 2006Pradier et al., 2011van den Akker et al., 2013.
Cell fusion
After infarction, grafted stem cells reduce cardiomyocyte apoptosis and stimulate cell proliferation via cell fusion Alvarez-Dolado et al., 2003Yang et al., 2012. Studies of bone marrow transplantation have proven that there is cell fusion between donor cells and recipient cells to form multi-nuclear cells Alvarez-Dolado et al., 2003Mayourian et al., 2016. Some studies have shown that stem cell transplantation can reprogram recipient cells, causing cardiomyocytes to re-enter the cell cycle; this contributes to regeneration of cardiac muscle and improvement of cardiac function Yeh and Zhang, 2006.
Manuel Alvarez-Dolado et al. demonstrated that bone marrow stromal cells (BMSCs) derived from R26R mice (i.e. a Cre reporter mouse line) could fuse with Cre+ neurosphere cells after 4 days of co-cuture. Moreover, BMSCs could fuse with Cre+ fibroblasts in primary cuture. In vivo studies have revealed that bone marrow of actin-Cre-GFP mice could be grafted to irradiated R26R mice. The transplanted cells were shown to fuse with and exhibit similar morphology as local mature cardiomyocytes. The fused cells expressed GFP at 2 months after implantation; however, GFP expression was absent in most of the fused cells at 4 months after implantation, suggesting that interesting changes may occur inside the cells Alvarez-Dolado et al., 2003. In accordance with Manuel et al., Yang et al. reported that transplanted bone marrow cells tend to integrate with local heart cells in infarcted regions rather then in healthy tissues. The infusion contributed to prevention of apoptosis of intergrated cardiomyocytes Yang et al., 2012. In another interesting study, from Nygren et al., it was shown that transplanted HSC survive but do not transdifferentiate into cardiomyocytes within the infarcted myocardium, whereas X-gal and GFP positive cardiomyocytes or fused cardiomyocytes are seen outside the infarcted zone Nygren et al., 2004.
Differentiation of transplanted cells into heart cells Transplantation of stem cells or progenitor cells restore structure and function of heart tissue after infarction; this has been demonstrated both by preclinical studies and clinical trials. One of the proposed mechanisms is that transplanted cells and mobilized cells have the ability to differentiate into heart cells to replace damaged or necrotic cells Kajstura et al., 2005Nagata et al., 2016Suzuki et al., 2015, possibly forming links with neighboring heart cells Dimmeler et al., 2005.
So far, stem cell differentiation into heart muscle cells is proposed to be related to four major signaling pathways: canonical/non-canonical Wnt signaling pathway, bone morphogenetic protein (BMP) signaling pathway, fibroblast growth factor (FGF) signaling pathway, and Notch signaling pathway. Firstly, when the canonical Wnt pathway is inhibited through Wnt3A and b-catenin inhibition, this stimulates the differentiation of stem cells/precursor cells into cardiomyocytes Pagliari et al., 2014. In addition, when non-canonical Wnt signaling pathway is activated through Wnt11 and Wnt5A activation, this also increases cell differentiation into cardiomyocytes Pagliari et al., 2014. Secondly, BMP-Smad1 inhibits Wnt/b-catenin and activates non-canonical Smad binding factors, leading to the transcription of activating transcription factor-2 (ATF2), while promoting major histocompatibility complex b (b-MHC) expression, contributing to heart cell differentiation Parikh et al., 2015. Thirdly, FGF activates the PI3K/Akt signaling pathway to preserve stem cell properties Parikh et al., 2015; moreover, via the MAPK/ERK signaling pathway they cause proliferation of cardiac progenitor cells but inhibition of their final differentiation into mature cardiomyocytes Tirosh-Finkel et al., 2010. Repression of FGF signaling, therefore, accelerates the differentiation process of cardiac precursor cells Tirosh-Finkel et al., 2010. Finally, Notch signaling plays a very important role in the regulation of stem cell differentiation into cardiomyocytes. It affects many kinds of cells- from transplanted cells, such as EPCs, MSCs and CPCs, to resident CSCs, immature cardiomyocytes and quiescent cardiomyocytes. Notch signaling is mediated by Jagged 1, NICD, Wnts, cyclin D1, RBP-Jk and Nkx2.5 Gude et al., 2015Luxan et al., 2016Zhou and Liu, 2014.
Arrhythmia
Besides tumorigenesis, the potential of arrhythmia induction from transplanted cells has also received wide attention. In clinical trials, it was shown that transplant of myoblasts could led to arrhythmia occurrence Leobon et al., 2003. Additionally, a number of other studies also confirmed that transplanted beating heart cells could give rise to arrhythmia Gillum and Sarvazyan, 2008Menasche et al., 2008, while transplantation of human ESC (hESC)-derived cardiovascular progenitors into human fetal hearts showed harmony of the structure and function between transplanted cells and cardiac host cells Ardehali et al., 2013. Moreover, the electromechanical coupling of hESC-derived cardiomyocytes and the suppression of cardiac arrhythmias in transplanted pig models seems to be related to the formation of electrical conduction bridges through scar tissues Shiba et al., 2012. The problem of arrhythmia after transplantation has now become a major problem, attracting scientists and clinicians to investigate mechanisms to overcome it.
Conclusion
In conclusion, transplanted cells delivered by several different pathways have shown that they could recover the structure and function of the damaged heart. These mechanisms include secretion of factors that protect heart cells, neovascularization, prevention of the fibrosis process, limiting the inflammatory processes, and mobilizing resident stem cells to repair the heart (by fusing with host cells or differentiating into heart cells for cell loss replacement). However, in addition to positive effects, transplanted cells can also have undesirable roles, such as induction of tumorigenesis or arrhythmia. The chosen cell type for transplantation needs to be considered carefully before clinical application.
Abbreviations
AKT/PKB: serine/threonine-specific protein kinase/ protein kinase B; ATF2: activating transcription factor-2; b-MHC: major histocompatibility complex b; Bad: Bcl-2-associated death promoter; Bcl-2: B-cell lymphoma 2 ; bFGF: basic fibroblast growth factor; BMMNCs: bone marrow monocytes; BMP: bone morphogenetic protein; BMSCs: bone marrow stromal cells; Ccl-2: chemokine (C-C motif) ligand 2; cGMP: cyclic guanosine monophosphate; CPCs: cardiac progenitor cells; CPCs: cardiac progenitor cells; CSCs: cardiac stem cells ; Cx43: mitochondrial connexin 43; CXCR4: C-X-C chemokine receptor type 4; DAG: phospholipid diacyl glycerol ; ECM: extracellular matrix; Ecs: endothelial cells; eNOS: endothelial nitric oxide synthase; EPCs: endothelial progenitor cells; EPDCs: epicardium-derived cells; EPO: erythropoietin; ESC-CM: embryonic stem cell cardiomyocyte; FGF: fibroblast growth factor; G-CSF: granulocyte colony stimulating factor; GATA4: GATA Binding Protein 4; GFP: green fluorescent protein; GSK-3: Glycogen synthase kinase 3; GSK-3: glycogen synthase kinase 3 beta; hESC: human embryonic stem cell; HGF: hepatocyte growth factor; HSCs: hematopoietic stem cells; IDO: indoleamine pyrrole 2,3-dioxygenase; IGF-1: insulin-like growth factor-1; IL: interleukin; iPSCs: induced pluripotent stem cells; JAK/STAT: Janus kinase/ signal transducers and activators of transcription; JAK2: Janus-activated tyrosine kinase 2; LV: left ventricular; MAPK/ERK: Mitogen-Activated Protein Kinase/ extracellular signal–regulated kinases; MCP-1: monocyte chemoattractant protein 1; MI: myocardial infarction; mKATP: mitochondrial ATP-dependent potassium channel; MMP: matrix metalloproteinase; MPTP: mitochondrial permeability transition pore; MSC-CM: MSC cardiomyocyte; MSCs: mesenchymal stem cells; NF-kB: nuclear factor kappa-light-chainenhancer of activated B cells; NICD: Notch intracellular domain; NO: Nitric oxide; PDH: pyruvate dehydrogenase; PGE2: prostaglandin E2; PI3K: Phosphatidylinositol-4,5-bisphosphate 3-kinase; PKC-a: protein kinase C alpha; PKC-d: protein kinase C delta; PKC-e: protein kinase C epsilon; PKC-z: protein kinase C zeta; PKG: protein kinase G; RACK: Receptor for activated c-kinase; RNS: reactive nitrogen species; ROS: reactive oxygen species; SDF-1: stromal cell-derived factor 1; Sfrp2: frizzled related protein 2; sKATP: sarcolemmal KATP; SMCs: smooth muscle cells; TB4: thymosin 4; TGF-beta: transforming growth factor-beta; TIMP-1: tissue inhibitor of metalloproteinase-1; TNF: tumor necrosis factor; VEGF: vascular endothelial growth factor; VSMCs: vascular smooth muscle cells; Wnt3a: Wnt Family Member 3A
Author Contributions
Truc Le-Buu Pham wrote and participated in editing the review. Phuc Van Pham oriented, gave important idea and revised the manuscript of this review.
References
-
S.G.
Almalki,
D.K.
Agrawal.
Effects of matrix metalloproteinases on the fate of mesenchymal stem cells. Stem cell research & therapy.
2016;
7
:
129
.
-
M.
Alvarez-Dolado,
R.
Pardal,
J.M.
Garcia-Verdugo,
J.R.
Fike,
H.O.
Lee,
K. Lois
Pfeffer,
S.J.
Morrison,
A.
Alvarez-Buylla.
Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature.
2003;
425
:
968-973
.
-
R.
Ardehali,
S.R.
Ali,
M.A.
Inlay,
O.J.
Abilez,
M.Q.
Chen,
T.A.
Blauwkamp,
M.
Yazawa,
Y.
Gong,
R.
Nusse,
M.
Drukker.
Prospective isolation of human embryonic stem cell-derived cardiovascular progenitors that integrate into human fetal heart tissue. Proc Natl Acad Sci U S A.
2013;
110
:
3405-3410
.
-
E.
Avolio,
M.
Meloni,
H.L.
Spencer,
F.
Riu,
R.
Katare,
G.
Mangialardi,
A.
Oikawa,
I.
Rodriguez-Arabaolaza,
Z.
Dang,
K.
Mitchell.
Combined intramyocardial delivery of human pericytes and cardiac stem cells additively improves the healing of mouse infarcted hearts through stimulation of vascular and muscular repair. Circ Res.
2015;
116
:
e81-94
.
-
A.
Azouz,
M.S.
Razzaque,
M.
El-Hallak,
T.
Taguchi.
Immunoinflammatory responses and fibrogenesis. Med Electron Microsc.
2004;
37
:
141-148
.
-
C.P.
Baines,
J.
Zhang,
G.W.
Wang,
Y.T.
Zheng,
J.X.
Xiu,
E.M.
Cardwell,
R.
Bolli,
P.
Ping.
Mitochondrial PKCepsilon and MAPK form signaling modules in the murine heart: enhanced mitochondrial PKCepsilon-MAPK interactions and differential MAPK activation in PKCepsilon-induced cardioprotection. Circ Res.
2002;
90
:
390-397
.
-
N.A.
Bax,
M.H.
van Marion,
B.
Shah,
M.-J.
Goumans,
C.V.
Bouten,
D.W.
van der Schaft.
Matrix production and remodeling capacity of cardiomyocyte progenitor cells during in vitro differentiation. Journal of molecular and cellular cardiology.
2012;
53
:
497-508
.
-
C.
Bearzi,
A.
Leri,
F.L.
Monaco,
M.
Rota,
A.
Gonzalez,
T.
Hosoda,
M.
Pepe,
K.
Qanud,
C.
Ojaimi,
S.
Bardelli.
Identification of a coronary vascular progenitor cell in the human heart. Proceedings of the National Academy of Sciences.
2009;
106
:
15885-15890
.
-
C.
Bearzi,
M.
Rota,
T.
Hosoda,
J.
Tillmanns,
A.
Nascimbene,
A.
De Angelis,
S.
YasuzawaAmano,
I.
Trofimova,
R.W.
Siggins,
N.
LeCapitaine.
Human cardiac stem cells. Proceedings of the National Academy of Sciences.
2007;
104
:
14068-14073
.
-
A.
Behfar,
L.V.
Zingman,
D.M.
Hodgson,
J.M.
Rauzier,
G.C.
Kane,
A.
Terzic,
M.
Puceat.
Stem cell differentiation requires a paracrine pathway in the heart. FASEB J.
2002;
16
:
1558-1566
.
-
A.P.
Beltrami,
L.
Barlucchi,
D.
Torella,
M.
Baker,
F.
Limana,
S.
Chimenti,
H.
Kasahara,
M.
Rota,
E.
Musso,
K.
Urbanek.
Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell.
2003;
114
:
763-776
.
-
U.
Ben-David,
N.
Benvenisty.
The tumorigenicity of human embryonic and induced pluripotent stem cells. Nature reviews Cancer.
2011;
11
:
268-277
.
-
T.
Ben-Mordechai,
R.
Holbova,
N.
Landa-Rouben,
T.
Harel-Adar,
M.S.
Feinberg,
I.
Abd Elrahman,
G.
Blum,
F.H.
Epstein,
Z.
Silman,
S.
Cohen.
Macrophage subpopulations are essential for infarct repair with and without stem cell therapy. J Am Coll Cardiol.
2013;
62
:
1890-1901
.
-
M.E.
Bernardo,
W.E.
Fibbe.
Mesenchymal stromal cells: sensors and switchers of inflammation. Cell Stem Cell.
2013;
13
:
392-402
.
-
M.F.
Berry,
A.J.
Engler,
Y.J.
Woo,
T.J.
Pirolli,
L.T.
Bish,
V.
Jayasankar,
K.J.
Morine,
T.J.
Gardner,
D.E.
Discher,
H.L.
Sweeney.
Mesenchymal stem cell injection after myocardial infarction improves myocardial compliance. Am J Physiol Heart Circ Physiol.
2006;
290
:
H2196-2203
.
-
S.
Bian,
L.
Zhang,
L.
Duan,
X.
Wang,
Y.
Min,
H.
Yu.
Extracellular vesicles derived from human bone marrow mesenchymal stem cells promote angiogenesis in a rat myocardial infarction model. Journal of Molecular Medicine.
2014;
92
:
387-397
.
-
G.
Blin,
D.
Nury,
S.
Stefanovic,
T.
Neri,
O.
Guillevic,
B.
Brinon,
V.
Bellamy,
C.
RuckerMartin,
P.
Barbry,
A.
Bel.
A purified population of multipotent cardiovascular progenitors derived from primate pluripotent stem cells engrafts in postmyocardial infarcted nonhuman primates. The Journal of clinical investigation.
2010;
120
:
1125-1139
.
-
K.M.
Broughton,
M.A.
Sussman.
Empowering Adult Stem Cells for Myocardial Regeneration V2.0: Success in Small Steps. Circ Res.
2016;
118
:
867-880
.
-
G.R.
Budas,
E.N.
Churchill,
D.
Mochly-Rosen.
Cardioprotective mechanisms of PKC isozyme-selective activators and inhibitors in the treatment of ischemiareperfusion injury. Pharmacological research.
2007;
55
:
523-536
.
-
T.
Cantz,
U.
Martin.
Induced pluripotent stem cells: characteristics and perspectives. Adv Biochem Eng Biotechnol.
2010;
123
:
107-126
.
-
C.
Cochain,
K.M.
Channon,
J.
Silvestre.
Angiogenesis in the Infarcted Myocardium. Antioxidants & Redox Signaling.
2013;
18
:
1100-1113
.
-
G.
Condorelli,
U.
Borello,
L.
De Angelis,
M.
Latronico,
D.
Sirabella,
M.
Coletta,
R.
Galli,
G.
Balconi,
A.
Follenzi,
G.
Frati.
Cardiomyocytes induce endothelial cells to trans-differentiate into cardiac muscle: implications for myocardium regeneration. Proc Natl Acad Sci U S A.
2001;
98
:
10733-10738
.
-
J.M.
Cregg,
M.A.
DePaul,
A.R.
Filous,
B.T.
Lang,
A.
Tran,
J.
Silver.
Functional regeneration beyond the glial scar. Exp Neurol.
2014;
253
:
197-207
.
-
Y.
Dai,
M.
Ashraf,
S.
Zuo,
R.
Uemura,
Y.S.
Dai,
Y.
Wang,
H.
Haider,
T.
Li,
M.
Xu.
Mobilized bone marrow progenitor cells serve as donors of cytoprotective genes for cardiac repair. Journal of molecular and cellular cardiology.
2008;
44
:
607-617
.
-
A.
Deb,
B.H.
Davis,
J.
Guo,
A.
Ni,
J.
Huang,
Z.
Zhang,
H.
Mu,
V.J.
Dzau.
SFRP2 regulates cardiomyogenic differentiation by inhibiting a positive transcriptional autofeedback loop of Wnt3a. Stem Cells.
2008;
26
:
35-44
.
-
S.
Dimmeler,
A.M.
Zeiher,
M.D.
Schneider.
Unchain my heart: the scientific foundations of cardiac repair. The Journal of clinical investigation.
2005;
115
:
572-583
.
-
J.A.
Dixon,
R.C.
Gorman,
R.E.
Stroud,
S.
Bouges,
H.
Hirotsugu,
J.H.
Gorman,
Martens
3rd,
S.
Itescu,
M.D.
Schuster,
T.
Plappert.
Mesenchymal cell transplantation and myocardial remodeling after myocardial infarction. Circulation.
2009;
120
:
S220-229
.
-
M.J.
Doyle,
T.J.
Maher,
Q.
Li,
M.G.
Garry,
B.P.
Sorrentino,
C.M.
Martin.
Abcg2-Labeled Cells Contribute to Different Cell Populations in the Embryonic and Adult Heart. Stem Cells Dev.
2016;
25
:
277-284
.
-
Y.Y.
Du,
S.H.
Zhou,
T.
Zhou,
H.
Su,
H.W.
Pan,
W.H.
Du,
B.
Liu,
Q.M.
Liu.
Immuno-inflammatory regulation effect of mesenchymal stem cell transplantation in a rat model of myocardial infarction. Cytotherapy.
2008;
10
:
469-478
.
-
S.S.
Fazel,
L.
Chen,
D.
Angoulvant,
S.H.
Li,
R.D.
Weisel,
A.
Keating,
R.K.
Li.
Activation of c-kit is necessary for mobilization of reparative bone marrow progenitor cells in response to cardiac injury. FASEB J.
2008;
22
:
930-940
.
-
P.
Ferdinandy,
R.
Schulz,
G.F.
Baxter.
Interaction of cardiovascular risk factors with myocardial ischemia/reperfusion injury, preconditioning, and postconditioning. Pharmacological reviews.
2007;
59
:
418-458
.
-
F.
Fernandez-Aviles,
J.A.
San Roman,
J.
Garcia-Frade,
M.E.
Fernandez,
M.J.
Penarrubia,
L.
de la Fuente,
M.
Gomez-Bueno,
A.
Cantalapiedra,
J.
Fernandez,
O.
Gutierrez.
Experimental and clinical regenerative capability of human bone marrow cells after myocardial infarction. Circ Res.
2004;
95
:
742-748
.
-
N.G.
Frangogiannis.
Regulation of the inflammatory response in cardiac repair. Circ Res.
2012;
110
:
159-173
.
-
N.G.
Frangogiannis,
C.W.
Smith,
M.L.
Entman.
The inflammatory response in myocardial infarction. Cardiovasc Res.
2002;
53
:
31-47
.
-
K.
Fuse,
M.
Kodama,
H.
Hanawa,
Y.
Okura,
M.
Ito,
T.
Shiono,
S.
Maruyama,
S.
Hirono,
K.
Kato,
K.
Watanabe.
Enhanced expression and production of monocyte chemoattractant protein-1 in myocarditis. Clinical and experimental immunology.
2001;
124
:
346-352
.
-
D.
Galli,
A.
Innocenzi,
L.
Staszewsky,
L.
Zanetta,
M.
Sampaolesi,
A.
Bai,
E.
Martinoli,
E.
Carlo,
G.
Balconi,
F.
Fiordaliso.
Mesoangioblasts, vessel-associated multipotent stem cells, repair the infarcted heart by multiple cellular mechanisms: a comparison with bone marrow progenitors, fibroblasts, and endothelial cells. Arterioscler Thromb Vasc Biol.
2005;
25
:
692-697
.
-
J.C.
Garbern,
R.T.
Lee.
Cardiac stem cell therapy and the promise of heart regeneration. Cell Stem Cell.
2013;
12
:
689-698
.
-
A.
Ghodsizad,
A.
Ruhparwar,
V.
Bordel,
E.
Mirsaidighazi,
H.M.
Klein,
M.M.
Koerner,
M.
Karck,
A.
El-Banayosy.
Clinical application of adult stem cells for therapy for cardiac disease. Cardiovascular therapeutics.
2013;
31
:
323-334
.
-
N.
Gillum,
N.
Sarvazyan.
Adhesion proteins, stem cells, and arrhythmogenesis. Cardiovascular toxicology.
2008;
8
:
1-13
.
-
S.
Glennie,
I.
Soeiro,
P.J.
Dyson,
E.W.
Lam,
F.
Dazzi.
Bone marrow mesenchymal stem cells induce division arrest anergy of activated T cells. Blood.
2005;
105
:
2821-2827
.
-
M.
Gnecchi,
H.
He,
N.
Noiseux,
O.D.
Liang,
L.
Zhang,
F.
Morello,
H.
Mu,
L.G.
Melo,
R.E.
Pratt,
J.S.
Ingwall.
Evidence supporting paracrine hypothesis for Aktmodified mesenchymal stem cell-mediated cardiac protection and functional improvement. Faseb j.
2006;
20
:
661-669
.
-
G.
Gómez-Mauricio,
I.
Moscoso,
M.-F.
Martín-Cancho,
V.
Crisóstomo,
C.
Prat-Vidal,
C.
Báez-Díaz,
F.M.
Sánchez-Margallo,
A.
Bernad.
Combined administration of mesenchymal stem cells overexpressing IGF-1 and HGF enhances neovascularization but moderately improves cardiac regeneration in a porcine model. Stem cell research & therapy.
2016;
7
:
94
.
-
N.
Gude,
E.
Joyo,
H.
Toko,
P.
Quijada,
M.
Villanueva,
N.
Hariharan,
V.
Sacchi,
S.
Truffa,
A.
Joyo,
M.
Voelkers.
Notch activation enhances lineage commitment and protective signaling in cardiac progenitor cells. Basic Res Cardiol.
2015;
110
:
29
.
-
S.
Harada,
Y.
Nakamura,
S.
Shiraya,
Y.
Fujiwara,
Y.
Kishimoto,
T.
Onohara,
Y.
Otsuki,
S.
Kishimoto,
Y.
Yamamoto,
I.
Hisatome.
Smooth muscle cell sheet transplantation preserve cardiac function and minimize cardiac remodeling in a rat myocardial infarction model. J Cardiothorac Surg.
2016;
11
:
131
.
-
N.
Hattan,
H.
Kawaguchi,
K.
Ando,
E.
Kuwabara,
J.
Fujita,
M.
Murata,
M.
Suematsu,
H.
Mori,
K.
Fukuda.
Purified cardiomyocytes from bone marrow mesenchymal stem cells produce stable intracardiac grafts in mice. Cardiovasc Res.
2005;
65
:
334-344
.
-
T.
Hosoda,
D.
D'Amario,
M.C.
Cabral-Da-Silva,
H.
Zheng,
M.E.
Padin-Iruegas,
B.
Ogorek,
J.
Ferreira-Martins,
S.
Yasuzawa-Amano,
K.
Amano,
N.
Ide-Iwata.
Clonality of mouse and human cardiomyogenesis in vivo. Proceedings of the National Academy of Sciences.
2009;
106
:
17169-17174
.
-
S.L.
House,
S.J.
Melhorn,
G.
Newman,
T.
Doetschman,
J.
Schultz Jel.
The protein kinase C pathway mediates cardioprotection induced by cardiac-specific overexpression of fibroblast growth factor-2. Am J Physiol Heart Circ Physiol.
2007;
293
:
H354-365
.
-
W.
Huang,
T.
Wang,
D.
Zhang,
T.
Zhao,
B.
Dai,
A.
Ashraf,
X.
Wang,
M.
Xu,
R.W.
Millard,
G.-C.
Fan.
Mesenchymal stem cells overexpressing CXCR4 attenuate remodeling of postmyocardial infarction by releasing matrix metalloproteinase-9. Stem cells and development.
2011;
21
:
778-789
.
-
I.
Huber,
I.
Itzhaki,
O.
Caspi,
G.
Arbel,
M.
Tzukerman,
A.
Gepstein,
M.
Habib,
L.
Yankelson,
I.
Kehat,
L.
Gepstein.
Identification and selection of cardiomyocytes during human embryonic stem cell differentiation. Faseb j.
2007;
21
:
2551-2563
.
-
S.
Jiang,
H.
Haider,
N.M.
Idris,
A.
Salim,
M.
Ashraf.
Supportive interaction between cell survival signaling and angiocompetent factors enhances donor cell survival and promotes angiomyogenesis for cardiac repair. Circ Res.
2006;
99
:
776-784
.
-
J.
Kajstura,
M.
Rota,
B.
Whang,
S.
Cascapera,
T.
Hosoda,
C.
Bearzi,
D.
Nurzynska,
H.
Kasahara,
E.
Zias,
M.
Bonafe.
Bone marrow cells differentiate in cardiac cell lineages after infarction independently of cell fusion. Circ Res.
2005;
96
:
127-137
.
-
H.
Kanazawa,
E.
Tseliou,
K.
Malliaras,
K.
Yee,
J.F.
Dawkins,
G.
De Couto,
R.R.
Smith,
M.
Kreke,
J.
Seinfeld,
R.C.
Middleton.
Cellular postconditioning: allogeneic cardiosphere-derived cells reduce infarct size and attenuate microvascular obstruction when administered after reperfusion in pigs with acute myocardial infarction. Circ Heart Fail.
2015;
8
:
322-332
.
-
K.
Kang,
R.
Ma,
W.
Cai,
W.
Huang,
C.
Paul,
J.
Liang,
Y.
Wang,
T.
Zhao,
H.W.
Kim,
M.
Xu.
Exosomes Secreted from CXCR4 Overexpressing Mesenchymal Stem Cells Promote Cardioprotection via Akt Signaling Pathway following Myocardial Infarction. Stem Cells Int.
2015;
2015
:
659890
.
-
V.
Karantalis,
W.
Balkan,
I.H.
Schulman,
K.E.
Hatzistergos,
J.M.
Hare.
Cellbased therapy for prevention and reversal of myocardial remodeling. Am J Physiol Heart Circ Physiol.
2012;
303
:
H256-270
.
-
D.H.
Kim,
Smith
Kshitiz,
P.
Kim,
E.H.
Ahn,
H.N.
Kim,
E.
Marban,
K.Y.
Suh,
A.
Levchenko.
Nanopatterned cardiac cell patches promote stem cell niche formation and myocardial regeneration. Integrative biology : quantitative biosciences from nano to macro.
2012;
4
:
1019-1033
.
-
P.J.
Kim,
M.
Mahmoudi,
X.
Ge,
Y.
Matsuura,
I.
Toma,
S.
Metzler,
N.G.
Kooreman,
J.
Ramunas,
C.
Holbrook,
M.V.
McConnell.
Direct evaluation of myocardial viability and stem cell engraftment demonstrates salvage of the injured myocardium. Circ Res.
2015;
116
:
e40-50
.
-
T.
Kinnaird,
E.
Stabile,
M.S.
Burnett,
C.W.
Lee,
S.
Barr,
S.
Fuchs,
S.E.
Epstein.
Marrow-derived stromal cells express genes encoding a broad spectrum of arteriogenic cytokines and promote in vitro and in vivo arteriogenesis through paracrine mechanisms. Circ Res.
2004;
94
:
678-685
.
-
A.A.
Kocher,
M.D.
Schuster,
M.J.
Szabolcs,
S.
Takuma,
D.
Burkhoff,
J.
Wang,
S.
Homma,
N.M.
Edwards,
S.
Itescu.
Neovascularization of ischemic myocardium by human bone-marrow-derived angioblasts prevents cardiomyocyte apoptosis, reduces remodeling and improves cardiac function. Nat Med.
2001;
7
:
430-436
.
-
C.
Kubal,
K.
Sheth,
B.
Nadal-Ginard,
M.
Galinanes.
Bone marrow cells have a potent anti-ischemic effect against myocardial cell death in humans. The Journal of thoracic and cardiovascular surgery.
2006;
132
:
1112-1118
.
-
A.J.
Leblanc,
Q.T.
Nguyen,
J.S.
Touroo,
A.L.
Aird,
R.C.
Chang,
C.K.
Ng,
J.B.
Hoying,
S.K.
Williams.
Adipose-derived cell construct stabilizes heart function and increases microvascular perfusion in an established infarct. Stem cells translational medicine.
2013;
2
:
896-905
.
-
A.S.
Lee,
C.
Tang,
M.S.
Rao,
I.L.
Weissman,
J.C.
Wu.
Tumorigenicity as a Clinical Hurdle for Pluripotent Stem Cell Therapies. Nat Med.
2013;
19
:
998-1004
.
-
B.
Leobon,
I.
Garcin,
P.
Menasche,
J.T.
Vilquin,
E.
Audinat,
S.
Charpak.
Myoblasts transplanted into rat infarcted myocardium are functionally isolated from their host. Proc Natl Acad Sci U S A.
2003;
100
:
7808-7811
.
-
J.
Leor,
S.
Gerecht,
S.
Cohen,
L.
Miller,
R.
Holbova,
A.
Ziskind,
M.
Shachar,
M.S.
Feinberg,
E.
Guetta,
J.
Itskovitz-Eldor.
Human embryonic stem cell transplantation to repair the infarcted myocardium. Heart (British Cardiac Society).
2007;
93
:
1278-1284
.
-
R.D.
Levit,
N.
Landazuri,
E.A.
Phelps,
M.E.
Brown,
A.J.
Garcia,
M.E.
Davis,
G.
Joseph,
R.
Long,
S.A.
Safley,
J.D.
Suever.
Cellular encapsulation enhances cardiac repair. Journal of the American Heart Association.
2013;
2
:
e000367
.
-
T.S.
Li,
K.
Cheng,
K.
Malliaras,
R.R.
Smith,
Y.
Zhang,
B.
Sun,
N.
Matsushita,
A.
Blusztajn,
J.
Terrovitis,
H.
Kusuoka.
Direct comparison of different stem cell types and subpopulations reveals superior paracrine potency and myocardial repair efficacy with cardiosphere-derived cells. J Am Coll Cardiol.
2012;
59
:
942-953
.
-
A.
Linke,
P.
Müller,
D.
Nurzynska,
C.
Casarsa,
D.
Torella,
A.
Nascimbene,
C.
Castaldo,
S.
Cascapera,
M.
Böhm,
F.
Quaini.
Stem cells in the dog heart are selfrenewing, clonogenic, and multipotent and regenerate infarcted myocardium, improving cardiac function. Proceedings of the National Academy of Sciences of the United States of America.
2005;
102
:
8966-8971
.
-
F.S.
Loffredo,
M.L.
Steinhauser,
J.
Gannon,
R.T.
Lee.
Bone marrow-derived cell therapy stimulates endogenous cardiomyocyte progenitors and promotes cardiac repair. Cell Stem Cell.
2011;
8
:
389-398
.
-
S.F.
Louis,
P.
Zahradka.
Vascular smooth muscle cell motility: From migration to invasion. Experimental & Clinical Cardiology.
2010;
15
:
e75-85
.
-
G.
Luxan,
G.
D'Amato,
D.
MacGrogan,
J.L.
de la Pompa.
Endocardial Notch Signaling in Cardiac Development and Disease. Circ Res.
2016;
118
:
e1-e18
.
-
W.
Lv,
W.
Li,
X.
Xu,
H.
Jiang,
O.Y.
Bang.
Bone marrow mesenchymal stem cells transplantation promotes the release of endogenous erythropoietin after ischemic stroke. Neural Regeneration Research.
2015;
10
:
1265-1270
.
-
J.
Mayourian,
R.M.
Savizky,
E.A.
Sobie,
K.D.
Costa.
Modeling Electrophysiological Coupling and Fusion between Human Mesenchymal Stem Cells and Cardiomyocytes. PLoS computational biology.
2016;
12
:
e1005014
.
-
P.
Menasche,
O.
Alfieri,
S.
Janssens,
W.
McKenna,
H.
Reichenspurner,
L.
Trinquart,
J.T.
Vilquin,
J.P.
Marolleau,
B.
Seymour,
J.
Larghero.
The Myoblast Autologous Grafting in Ischemic Cardiomyopathy (MAGIC) trial: first randomized placebo-controlled study of myoblast transplantation. Circulation.
2008;
117
:
1189-1200
.
-
P.
Menasche,
A.A.
Hagege,
M.
Scorsin,
B.
Pouzet,
M.
Desnos,
D.
Duboc,
K.
Schwartz,
J.T.
Vilquin,
J.P.
Marolleau.
Myoblast transplantation for heart failure. Lancet.
2001;
357
:
279-280
.
-
C.
Mias,
O.
Lairez,
E.
Trouche,
J.
Roncalli,
D.
Calise,
M.H.
Seguelas,
C.
Ordener,
M.D.
Piercecchi-Marti,
N.
Auge,
A.N.
Salvayre.
Mesenchymal stem cells promote matrix metalloproteinase secretion by cardiac fibroblasts and reduce cardiac ventricular fibrosis after myocardial infarction. Stem Cells.
2009;
27
:
2734-2743
.
-
M.
Mirotsou,
Z.
Zhang,
A.
Deb,
L.
Zhang,
M.
Gnecchi,
N.
Noiseux,
H.
Mu,
A.
Pachori,
V.
Dzau.
Secreted frizzled related protein 2 (Sfrp2) is the key Aktmesenchymal stem cell-released paracrine factor mediating myocardial survival and repair. Proc Natl Acad Sci U S A.
2007;
104
:
1643-1648
.
-
T.
Nagai,
I.
Komuro.
Gene and cytokine therapy for heart failure: molecular mechanisms in the improvement of cardiac function. Am J Physiol Heart Circ Physiol.
2012;
303
:
H501-512
.
-
H.
Nagata,
M.
Ii,
E.
Kohbayashi,
M.
Hoshiga,
T.
Hanafusa,
M.
Asahi.
Cardiac Adipose-Derived Stem Cells Exhibit High Differentiation Potential to Cardiovascular Cells in C57BL/6 Mice. Stem cells translational medicine.
2016;
5
:
141-151
.
-
N.
Nagaya,
T.
Fujii,
T.
Iwase,
H.
Ohgushi,
T.
Itoh,
M.
Uematsu,
M.
Yamagishi,
H.
Mori,
K.
Kangawa,
S.
Kitamura.
Intravenous administration of mesenchymal stem cells improves cardiac function in rats with acute myocardial infarction through angiogenesis and myogenesis. Am J Physiol Heart Circ Physiol.
2004;
287
:
H2670-2676
.
-
N.
Nagaya,
K.
Kangawa,
T.
Itoh,
T.
Iwase,
S.
Murakami,
Y.
Miyahara,
T.
Fujii,
M.
Uematsu,
H.
Ohgushi,
M.
Yamagishi.
Transplantation of mesenchymal stem cells improves cardiac function in a rat model of dilated cardiomyopathy. Circulation.
2005;
112
:
1128-1135
.
-
A.J.
Nauta,
A.B.
Kruisselbrink,
E.
Lurvink,
R.
Willemze,
W.E.
Fibbe.
Mesenchymal stem cells inhibit generation and function of both CD34+-derived and monocyte-derived dendritic cells. Journal of immunology.
2006;
(Baltimore
:
Md : 1950) 177, 2080-2087
.
-
N.
Noiseux,
M.
Gnecchi,
M.
Lopez-Ilasaca,
L.
Zhang,
S.D.
Solomon,
A.
Deb,
V.J.
Dzau,
R.E.
Pratt.
Mesenchymal stem cells overexpressing Akt dramatically repair infarcted myocardium and improve cardiac function despite infrequent cellular fusion or differentiation. Mol Ther.
2006;
14
:
840-850
.
-
J.
Nussbaum,
E.
Minami,
M.A.
Laflamme,
J.A.
Virag,
C.B.
Ware,
A.
Masino,
V.
Muskheli,
L.
Pabon,
H.
Reinecke,
C.E.
Murry.
Transplantation of undifferentiated murine embryonic stem cells in the heart: teratoma formation and immune response. Faseb j.
2007;
21
:
1345-1357
.
-
J.M.
Nygren,
S.
Jovinge,
M.
Breitbach,
P.
Sawen,
W.
Roll,
J.
Hescheler,
J.
Taneera,
B.K.
Fleischmann,
S.E.
Jacobsen.
Bone marrow-derived hematopoietic cells generate cardiomyocytes at a low frequency through cell fusion, but not transdifferentiation. Nat Med.
2004;
10
:
494-501
.
-
S.
Ohnishi,
B.
Yanagawa,
K.
Tanaka,
Y.
Miyahara,
H.
Obata,
M.
Kataoka,
M.
Kodama,
H.
Ishibashi-Ueda,
K.
Kangawa,
S.
Kitamura.
Transplantation of mesenchymal stem cells attenuates myocardial injury and dysfunction in a rat model of acute myocarditis. Journal of molecular and cellular cardiology.
2007a;
42
:
88-97
.
-
S.
Ohnishi,
T.
Yasuda,
S.
Kitamura,
N.
Nagaya.
Effect of hypoxia on gene expression of bone marrow-derived mesenchymal stem cells and mononuclear cells. Stem Cells.
2007b;
25
:
1166-1177
.
-
K.
Okita,
M.
Nakagawa,
H.
Hyenjong,
T.
Ichisaka,
S.
Yamanaka.
Generation of mouse induced pluripotent stem cells without viral vectors. Science.
2008;
322
:
949-953
.
-
Y.
Onai,
J.
Suzuki,
Y.
Maejima,
G.
Haraguchi,
S.
Muto,
A.
Itai,
M.
Isobe.
Inhibition of NF-{kappa}B improves left ventricular remodeling and cardiac dysfunction after myocardial infarction. Am J Physiol Heart Circ Physiol.
2007;
292
:
H530-538
.
-
R.R.
Padua,
P.L.
Merle,
B.W.
Doble,
C.H.
Yu,
P.
Zahradka,
G.N.
Pierce,
V.
Panagia,
E.
Kardami.
FGF-2-induced negative inotropism and cardioprotection are inhibited by chelerythrine: involvement of sarcolemmal calcium-independent protein kinase C. Journal of molecular and cellular cardiology.
1998;
30
:
2695-2709
.
-
S.
Pagliari,
J.
Jelinek,
G.
Grassi,
G.
Forte.
Targeting pleiotropic signaling pathways to control adult cardiac stem cell fate and function. Frontiers in physiology.
2014;
5
:
219
.
-
A.
Parikh,
J.
Wu,
R.M.
Blanton,
E.S.
Tzanakakis.
Signaling Pathways and Gene Regulatory Networks in Cardiomyocyte Differentiation. Tissue engineering Part.
2015;
B
:
Reviews 21, 377-392
.
-
E.C.
Perin,
K.M.
Borow,
G.V.
Silva,
A.N.
DeMaria,
O.C.
Marroquin,
P.P.
Huang,
J.H.
Traverse,
H.
Krum,
D.
Skerrett,
Y.
Zheng.
A Phase II Dose-Escalation Study of Allogeneic Mesenchymal Precursor Cells in Patients With Ischemic or Nonischemic Heart Failure. Circ Res.
2015;
117
:
576-584
.
-
A.
Pradier,
J.
Passweg,
J.
Villard,
V.
Kindler.
Human bone marrow stromal cells and skin fibroblasts inhibit natural killer cell proliferation and cytotoxic activity. Cell transplantation.
2011;
20
:
681-691
.
-
M.
Ramalho-Santos,
S.
Yoon,
Y.
Matsuzaki,
R.C.
Mulligan,
D.A.
Melton.
"Stemness": transcriptional profiling of embryonic and adult stem cells. Science.
2002;
298
:
597-600
.
-
B.A.
Rose,
T.
Force,
Y.
Wang.
Mitogen-Activated Protein Kinase Signaling in the Heart: Angels Versus Demons in a Heart-Breaking Tale. Physiological reviews.
2010;
90
.
-
M.
Rota,
M.E.
Padin-Iruegas,
Y.
Misao,
A.
De Angelis,
S.
Maestroni,
J.
Ferreira-Martins,
E.
Fiumana,
R.
Rastaldo,
M.L.
Arcarese,
T.S.
Mitchell.
Local activation or implantation of cardiac progenitor cells rescues scarred infarcted myocardium improving cardiac function. Circulation research.
2008;
103
:
107-116
.
-
Y.
Shiba,
S.
Fernandes,
W.Z.
Zhu,
D.
Filice,
V.
Muskheli,
J.
Kim,
N.J.
Palpant,
J.
Gantz,
K.W.
Moyes,
H.
Reinecke.
Human ES-cell-derived cardiomyocytes electrically couple and suppress arrhythmias in injured hearts. Nature.
2012;
489
:
322-325
.
-
J.
Silver,
J.H.
Miller.
Regeneration beyond the glial scar. Nat Rev Neurosci.
2004;
5
:
146-156
.
-
N.
Smart,
K.N.
Dube,
P.R.
Riley.
Epicardial progenitor cells in cardiac regeneration and neovascularisation. Vascular pharmacology.
2013;
58
:
164-173
.
-
N.
Smart,
C.A.
Risebro,
A.A.
Melville,
K.
Moses,
R.J.
Schwartz,
K.R.
Chien,
P.R.
Riley.
Thymosin beta4 induces adult epicardial progenitor mobilization and neovascularization. Nature.
2007;
445
:
177-182
.
-
M.H.
Soonpaa,
G.Y.
Koh,
M.G.
Klug,
L.J.
Field.
Formation of nascent intercalated disks between grafted fetal cardiomyocytes and host myocardium. Science.
1994;
264
:
98-101
.
-
W.
Srisakuldee,
Z.
Makazan,
B.E.
Nickel,
F.
Zhang,
J.A.
Thliveris,
K.B.
Pasumarthi,
E.
Kardami.
The FGF-2-triggered protection of cardiac subsarcolemmal mitochondria from calcium overload is mitochondrial connexin 43-dependent. Cardiovasc Res.
2014;
103
:
72-80
.
-
E.
Suzuki,
D.
Fujita,
M.
Takahashi,
S.
Oba,
H.
Nishimatsu.
Adipose tissuederived stem cells as a therapeutic tool for cardiovascular disease. World journal of cardiology.
2015;
7
:
454-465
.
-
X.L.
Tang,
G.
Rokosh,
S.K.
Sanganalmath,
Y.
Tokita,
M.C.
Keith,
G.
Shirk,
H.
Stowers,
G.N.
Hunt,
W.
Wu,
B.
Dawn.
Effects of Intracoronary Infusion of Escalating Doses of Cardiac Stem Cells in Rats With Acute Myocardial Infarction. Circ Heart Fail.
2015;
8
:
757-765
.
-
J.
Tillmanns,
M.
Rota,
T.
Hosoda,
Y.
Misao,
G.
Esposito,
A.
Gonzalez,
S.
Vitale,
C.
Parolin,
S.
Yasuzawa-Amano,
J.
Muraski.
Formation of large coronary arteries by cardiac progenitor cells. Proceedings of the National Academy of Sciences.
2008;
105
:
1668-1673
.
-
L.
Tirosh-Finkel,
A.
Zeisel,
M.
Brodt-Ivenshitz,
A.
Shamai,
Z.
Yao,
R.
Seger,
E.
Domany,
E.
Tzahor.
BMP-mediated inhibition of FGF signaling promotes cardiomyocyte differentiation of anterior heart field progenitors. Development.
2010;
(Cambridge
:
England) 137, 2989-3000
.
-
S.
Tohyama,
F.
Hattori,
M.
Sano,
T.
Hishiki,
Y.
Nagahata,
T.
Matsuura,
H.
Hashimoto,
T.
Suzuki,
H.
Yamashita,
Y.
Satoh.
Distinct metabolic flow enables largescale purification of mouse and human pluripotent stem cell-derived cardiomyocytes. Cell Stem Cell.
2013;
12
:
127-137
.
-
K.
Urbanek,
D.
Cesselli,
M.
Rota,
A.
Nascimbene,
A.
De Angelis,
T.
Hosoda,
C.
Bearzi,
A.
Boni,
R.
Bolli,
J.
Kajstura.
Stem cell niches in the adult mouse heart. Proceedings of the National Academy of Sciences.
2006;
103
:
9226-9231
.
-
K.
Urbanek,
F.
Quaini,
G.
Tasca,
D.
Torella,
C.
Castaldo,
B.
Nadal-Ginard,
A.
Leri,
J.
Kajstura,
E.
Quaini,
P.
Anversa.
Intense myocyte formation from cardiac stem cells in human cardiac hypertrophy. Proceedings of the National Academy of Sciences.
2003;
100
:
10440-10445
.
-
C.
Urbich,
A.
Aicher,
C.
Heeschen,
E.
Dernbach,
W.K.
Hofmann,
A.M.
Zeiher,
S.
Dimmeler.
Soluble factors released by endothelial progenitor cells promote migration of endothelial cells and cardiac resident progenitor cells. Journal of molecular and cellular cardiology.
2005;
39
:
733-742
.
-
F.
Akker,
S.C.
de Jager,
J.P.
Sluijter.
Mesenchymal stem cell therapy for cardiac inflammation: immunomodulatory properties and the influence of toll-like receptors. Mediators of inflammation.
2013;
2013
:
181020
.
-
Y.
Wu,
J.E.
Ip,
J.
Huang,
L.
Zhang,
K.
Matsushita,
C.C.
Liew,
R.E.
Pratt,
V.J.
Dzau.
Essential role of ICAM-1/CD18 in mediating EPC recruitment, angiogenesis, and repair to the infarcted myocardium. Circ Res.
2006;
99
:
315-322
.
-
M.
Xu,
R.
Uemura,
Y.
Dai,
Y.
Wang,
Z.
Pasha,
M.
Ashraf.
In vitro and in vivo effects of bone marrow stem cells on cardiac structure and function. Journal of molecular and cellular cardiology.
2007;
42
:
441-448
.
-
X.
Xu,
Z.
Xu,
Y.
Xu,
G.
Cui.
Effects of mesenchymal stem cell transplantation on extracellular matrix after myocardial infarction in rats. Coronary artery disease.
2005;
16
:
245-255
.
-
Y.J.
Xu,
D.
Chapman,
I.M.
Dixon,
R.
Sethi,
X.
Guo,
N.S.
Dhalla.
Differential gene expression in infarct scar and viable myocardium from rat heart following coronary ligation. J Cell Mol Med.
2004;
8
:
85-92
.
-
W.J.
Yang,
S.H.
Li,
R.D.
Weisel,
S.M.
Liu,
R.K.
Li.
Cell fusion contributes to the rescue of apoptotic cardiomyocytes by bone marrow cells. J Cell Mol Med.
2012;
16
:
3085-3095
.
-
K.
Yee,
K.
Malliaras,
H.
Kanazawa,
E.
Tseliou,
K.
Cheng,
D.J.
Luthringer,
C.S.
Ho,
K.
Takayama,
N.
Minamino,
J.F.
Dawkins.
Allogeneic cardiospheres delivered via percutaneous transendocardial injection increase viable myocardium, decrease scar size, and attenuate cardiac dilatation in porcine ischemic cardiomyopathy. PLoS One.
2014;
9
:
e113805
.
-
E.T.
Yeh,
S.
Zhang.
A novel approach to studying transformation of human stem cells into cardiac cells in vivo. The Canadian journal of cardiology.
2006;
22 Suppl B
:
66b-71b
.
-
Y.S.
Yoon,
A.
Wecker,
L.
Heyd,
J.S.
Park,
T.
Tkebuchava,
K.
Kusano,
A.
Hanley,
H.
Scadova,
G.
Qin,
D.H.
Cha.
Clonally expanded novel multipotent stem cells from human bone marrow regenerate myocardium after myocardial infarction. The Journal of clinical investigation.
2005;
115
:
326-338
.
-
B.
Yu,
H.W.
Kim,
M.
Gong,
J.
Wang,
R.W.
Millard,
Y.
Wang,
M.
Ashraf,
M.
Xu.
Exosomes secreted from GATA-4 overexpressing mesenchymal stem cells serve as a reservoir of anti-apoptotic microRNAs for cardioprotection. Int J Cardiol.
2015;
182
:
349-360
.
-
R.
Zamilpa,
M.L.
Lindsey.
Extracellular matrix turnover and signaling during cardiac remodeling following MI: causes and consequences. Journal of molecular and cellular cardiology.
2010;
48
:
558-563
.
-
M.
Zhang,
D.
Methot,
V.
Poppa,
Y.
Fujio,
K.
Walsh,
C.E.
Murry.
Cardiomyocyte grafting for cardiac repair: graft cell death and anti-death strategies. Journal of molecular and cellular cardiology.
2001;
33
:
907-921
.
-
Z.
Zhang,
A.
Deb,
Z.
Zhang,
A.
Pachori,
W.
He,
J.
Guo,
R.
Pratt,
V.J.
Dzau.
Secreted frizzled related protein 2 protects cells from apoptosis by blocking the effect of canonical Wnt3a. Journal of molecular and cellular cardiology.
2009;
46
:
370-377
.
-
T.
Zhao,
W.
Zhao,
Y.
Chen,
R.A.
Ahokas,
Y.
Sun.
Vascular endothelial growth factor (VEGF)-A: role on cardiac angiogenesis following myocardial infarction. Microvascular research.
2010;
80
:
188-194
.
-
X.L.
Zhou,
J.C.
Liu.
Role of Notch signaling in the mammalian heart. Brazilian journal of medical and biological research = Revista brasileira de pesquisas medicas e biologicas / Sociedade Brasileira de Biofisica [et al].
2014;
47
:
1-10
.
Comments
Downloads
Article Details
Volume & Issue : Vol 3 No 11 (2016)
Page No.: 951-972
Published on: 2016-11-18
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
- HTML viewed - 5104 times
- Download PDF downloaded - 1398 times
- View Article downloaded - 58 times
 Biomedpress
Biomedpress