Abstract
Introduction: Motor-function modulation through Pyridine-3-carboxylic acid was assessed against. Haloperidol induced Parkinson’s disease (PD) in albino-mice. The objectives of this study were to test the effect of Haloperidol in development of PD, effectiveness of Pyridine-3-carboxylic acid in mice and evaluation of the motor-function changes in mice before and after treatment.
Methods: The study was divided into 3 phases: During Phase-I (randomization), all the subjects were randomly divided into 4 groups and trained for wire-hanging, grip strength, vertical rod and swim tests for 1 week. During Phase-II (disease induction), PD was induced by intra-peritoneal (ip) administration of Haloperidol (HP) in a dose of 1 mg/kg/d for 7 days. Group-A comprised of healthy controls, Group-B (Diseased), Group-C (HP+Pyridine-3-carboxylic acid 7.15 mg/kg/d) and Group-D (HP+Pyridine-3-carboxylic acid15 mg/kg/d).
Results: Pyridine-3-carboxylic acid treatment continued for 5 weeks. During Phase-III the above mention tests were performed and the effects of Pyridine-3-carboxylic acid were recorded. However, in swim test Group D is statistically insignificant as compared to Group B (p=0.284). In recent study, haloperidol is found to be effective in inducing motor function anomalies likewise in Parkinson’s disease including movement slowness, difficulties with gait and balance.
Conclusion: oral administration of Pyridine-3-carboxylic acid improved Motor-function changes in diseased mice. Therefore, it is concluded that Pyridine-3-carboxylic acid may help to alleviate PD symptoms.
Introduction
Parkinson’s disease (PD) was first illustrated by James Parkinson in 1817 in “An Essay on the Shaking Palsy” Kempster et al., 2007. The major symptoms of PD are tremors, bradykinesia, postural instability and rigidity, postural abnormalities, akinesia and festinating gait. These symptoms are led by psychological symptoms such as depression and more general non-motor symptoms such as olfactory dysfunction constipation, and sleep disturbances Klockgether, 2004. The incidence of Parkinson’s disease (PD) is nearly about 1% at the age of 65 years which further increased to 5% with the age of 85 years Hirtz et al., 2007. Haloperidol is a typical neuroleptic drug and shows effect by blocking the postsynaptic dopamine D2 receptors in the mesolimbic system and cause an escalation of dopamine turnover by blockage of the D2 receptors Zaidi et al., 2016a. Anticholinergic and β-adrenergic receptor blocking effects of haloperidol is quite weak. Parkinson disease is characterized majorly by the loss of melanin containing dopaminergic neurons in zona compacta of the substantia nigra Bernheimer et al., 1973. Haloperidol causes a decrease in dopamine neurotransmission Naidu et al., 2003. Haloperidol exerts its antipsychotic effect most likely through potent blockade of central dopamine receptors and marked rigidity linked with haloperidol administration Zaidi et al., 2016b. In animal studies, neurotoxins such as 1-methyl-4-phenyl-1,2,3,6-tetrahydroxypyridine (MPTP) and 6-OHDA and haloperidol are used commonly to create experimental model of PD Fernagut et al., 2002Sheidaei, 2010 by which certain aspects of the disease such as motor abnormalities and slowing and of movement and catalepsy can be modeled Scholtissen et al., 2006Wang et al., 2005. Clinical symptoms appear only when dopaminergic neuronal death exceeds a critical threshold 70-80% of striatal nerve terminals.
Vitamin B3 also known as Pyridine-3-carboxylic acid and found in foods including certain types of meat and organ meat, tuna fish, seeds, mushrooms and others. Vitamin B3, which is usually medically referred to as Pyridine-3-carboxylic acid, comes in 3 forms nicotinic acid, Pyridine-3-carboxylic acid amide and Inositol HexaPyridine-3-carboxylic acidate. Vitamin B3 Pyridine-3-carboxylic acid has been studied extensively to treat of many commonly occurring health problems. Pyridine-3-carboxylic acid is an important vitamin for maintaining healthy brain function and healthy cardiovascular system and metabolism, especially balancing blood cholesterol levels Gurakar et al., 1985Morris et al., 2004. Nicotinic acid (Pyridine-3-carboxylic acid) has shown neuro-protective role in mice-stroke model by promoting the monocyte polarization into protective phenotype in brain (Rahman et al., 2014). Absorbed niacin is used in the synthesis of nicotinamide adenine dinucleotide (NAD) in the body, and in the metabolic process NAD releases nicotinamide by poly ADP-ribosylation, the activation of which has been reported to mediate 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-induced Parkinson’s disease Molina et al., 1996.
The objectives of this study were to evaluate the effect of Haloperidol in development of PD, effectiveness of Pyridine-3-carboxylic acid to modulate Motor-function activities in mice model PD and record the Motor-function changes in mice before & after the treatment of PD with Pyridine-3-carboxylic acid.
Materials - Methods
Study design
Albino mice (20-25g) were taken from animal house of UVAS (University of veterinary sciences, Lahore-Pakistan). The animals were acclimatized and kept under specified temperature (22±2°C) and humidity (60±2%) under 12-hours light/dark cycles with food and water ad libitum. Experimental procedures and animal handling were approved by Institutional Committee of Research Ethics, Hajvery University (Ethical no. 720EN-2016) Lahore, Pakistan.
The study was divided into three phases. Table 1
Phase-I
During Phase-I, the motor-function modeling of mice was performed using wire hanging test, grip strength test, vertical rod test, and swim test. All subjects were trained and divided into four groups comprising of 7 mice in each.
Wire Hanging Test
The wire hanging test was used to assess muscle function and coordination over time. The test is based on the latency of a mouse to fall off a metal wire upon exhaustion. The wire hanging test is performed in order to demonstrate a motor neuromuscular impairment and motor coordination. This test was also used for evaluating the neuromuscular tone. A 55 cm wide 2 mm thick wire is secured to two vertical stands. The wire must be tightly attached to the frame to avoid vibration or unwanted displacement of the wire while the investigator is handling the animals or during the measurements, since these unwanted effects would interfere with the animal’s performance Aartsma-Rus and van Putten, 2014Klein and Lewis, 2012.
Grip Strength Test
The grip strength test is a modest non-intrusive method intended to assess mouse muscle power in vivo, by taking benefit of the animal's affinity to grip a flat metal bar or framework while suspended by its tail or each of the four appendages. This is a pure test of strength, although as for any test motivational factors could potentially play a role. The inverted screen is a 43 cm square of wire mesh consisting of 12 mm squares of 1 mm diameter wire. We place the mouse in the center of the wire mesh screen, start a stop clock, and rotate the screen to an inverted position over 2 sec, with the mouse's head declining first. We hold the screen steadily 40-50 cm above a padded surface.
Vertical Pole Test
Vertical pole test is used to measure the sensorimotor function of mice. PD motor tests provide a good read-out of neurological function.. Each mouse is placed head upwards at the centre of a round pole that is inclined at 90° and performance is determined by the latency(s) of the mouse to turn downwards and completely descend the pole. In a habituation period one day prior to testing, each mouse is allowed to attempt to descend the pole. Each testing session lasts for a maximum of 180 seconds.
Swim Test
The forced swim test, also known as the behavioral despair test, is used to test for depression-like behavior in both mice and rats. The test includes placing a rat or mouse inside a cylinder filled with water. The mobility of the animal is measured. Traditionally, ‘floating behavior’ (the animal remains almost immobile and with its head above water). The forced swim test is a mice Motor-function test utilized for assessment of stimulant medications, upper adequacy of new mixes, and trial controls that are gone for rendering or anticipating depressive-like states
Phase-II
During Phase-II, PD was induced by administering Haloperidol of 1mg / kg per day (ip) for 7 days. All animals were observed for 30 minutes post injection and then hourly intervals for next 3 hours. At the end of the 7th day, PD was assessed by hind limb movements and behavior Manikandaselvi et al., 2012. After induction of disease, subjects were divided into four groups. Group A (Normal) containing normal mice with (i.p) injection of saline served as control. Group B served as diseased group received (i.p) injection of haloperidol (1 mg/kg per day) for seven days. Group C and D were administered Pyridine-3-carboxylic acid orally, 7.15 mg and 15 mg per kg of body weight orally for 36 days respectively.
Phase-III
During Phase-III, following four tests were performed on daily basis to check the difference in behavior before induction of PD and after treatment. These tests include wire hanging test, grip strength test, Vertical rod test, and swim test Zaidi et al., 2016a.
Statistical Analysis
Data was statistically analyzed on SPSS version 22.0 using ANOVA with a p < 0.05 considered as significant.
Results
The present study was conducted to assess the neuro-protective and motor-function modulation activity of Pyridine-3-carboxylic acid in HP induced PD animal model. Mice were acclimatized and their Motor-function modeling was done. Following four tests were applied to mice. These are swim test, vertical pole, grip strength, and wire hanging test. During these tests, physical and social activities of the mice were monitored.
Wire Hanging Test
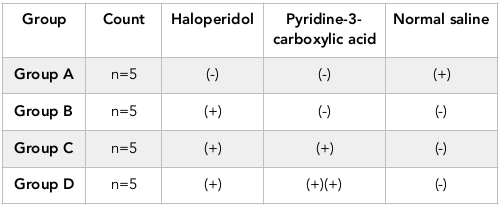
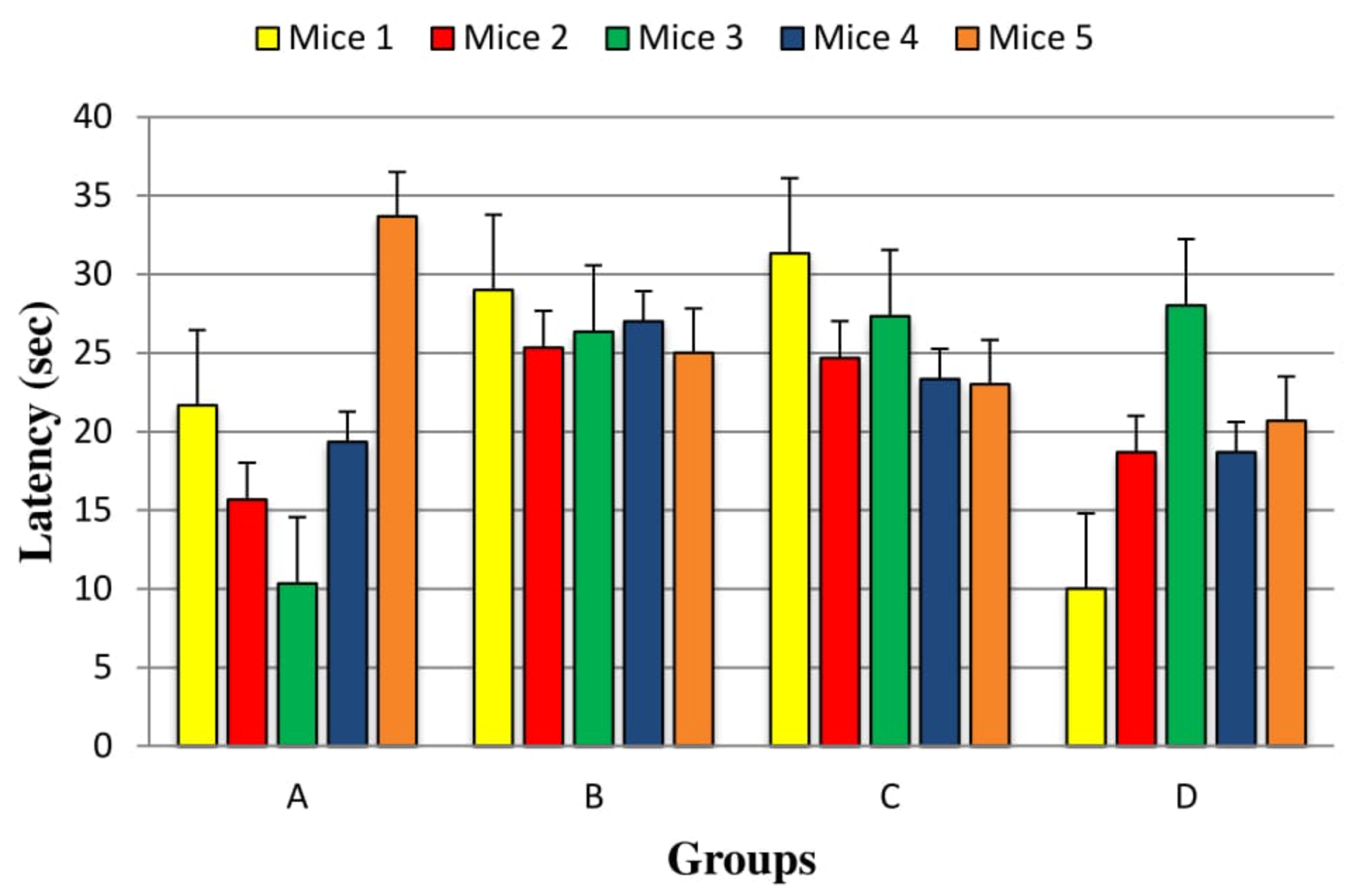
During motor-function modulation, an important parameter for treatment effectiveness is neuromuscular strength. Wire hanging test was performed to assess the neuromuscular ability of mice. While performing this test, animal were suspended by its fore paws with a 2 mm wire 30 cm above the ground. By performing this test, the measure of motor coordination and animal’s ability to take on its hind limbs and tail with a specific end goal to grasp wire was observed. Latency to fall was measured from the time a mouse hanged by its forepaws till it falls. The test was performed five times for each mouse and a mean value was considered and analyzed by one way ANOVA. It was observed during experimentation that Group D showed more neuromuscular strength than Group C as shown in Figure 1 . However, results of group D were not statistically much significant (P=0.082) in comparison with Group C.
Grip Strength Test
The modified screen is a 43 cm square of wire cross section comprising of 12 mm squares of 1 mm measurement wire. It is encompassed by a 4 cm profound wooden beading which keeps the infrequent mouse which endeavors to from hopping on to the next side. It is a test of muscle quality utilizing each of the four appendages. Most ordinary mice effortlessly score greatest on this undertaking; it is a brisk yet heartless gross screen. A common error with commercial strength meters is that the bar or other grip feature is not thin enough for mice to exert a maximum grip. As a general rule, the thinner the wire or bar, the better a mouse can grip with its small claws.
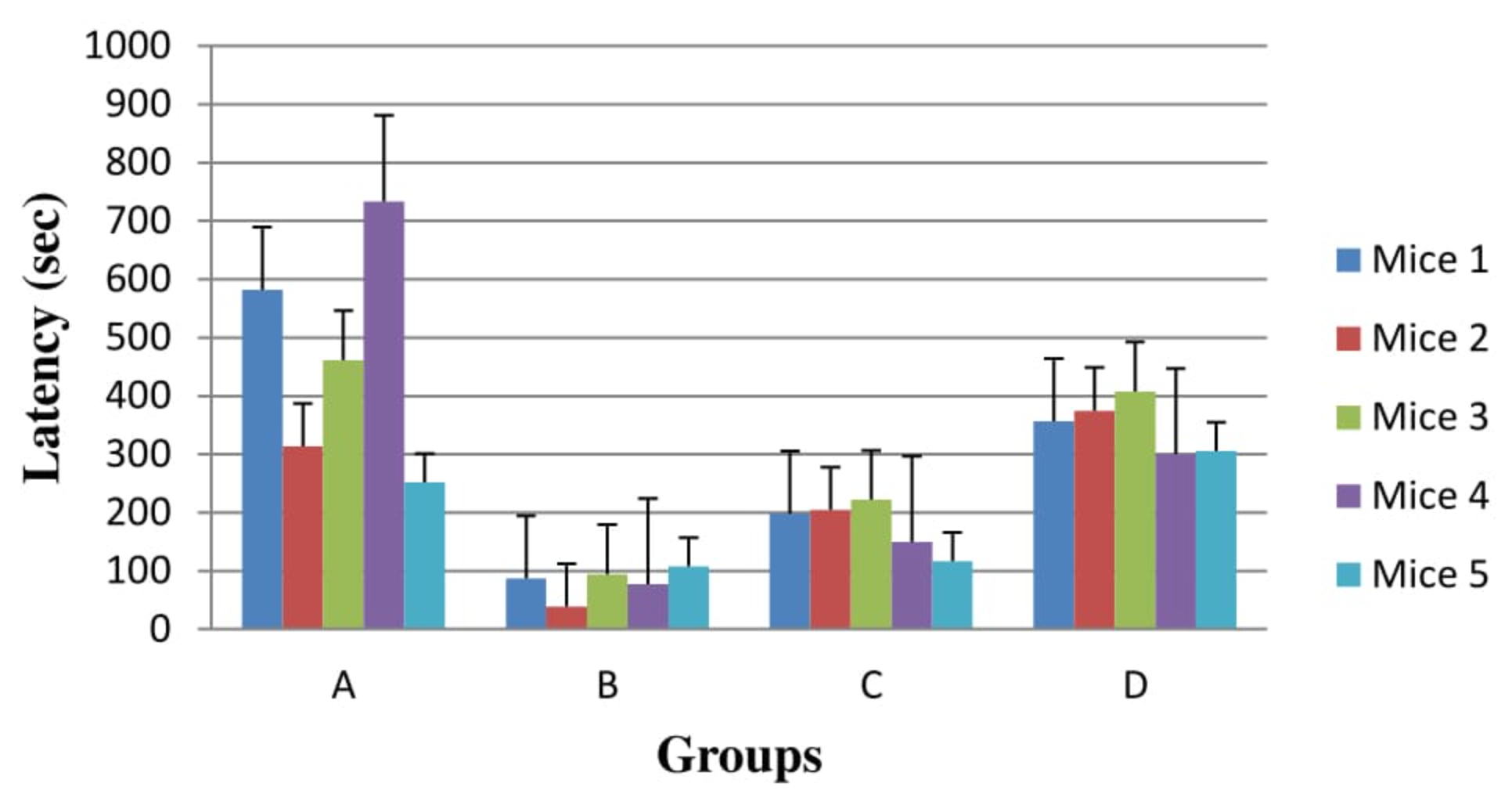
Another key feature for treatment effectiveness is neuromuscular strength. This is an immaculate test of quality, despite the fact that concerning any test motivational variables could conceivably assume a part. Grip strength test showed that Group D mice had strong grip as compared to Group C and Group B mice as shown in Figure 2 . Results were found statistically significant for Group D in comparison with Group C (P=0.047). It was also observed during study that Group B had shown least strength of grip than Group A (P=0.005) as demonstrated in Figure 2 .
Vertical Pole Test
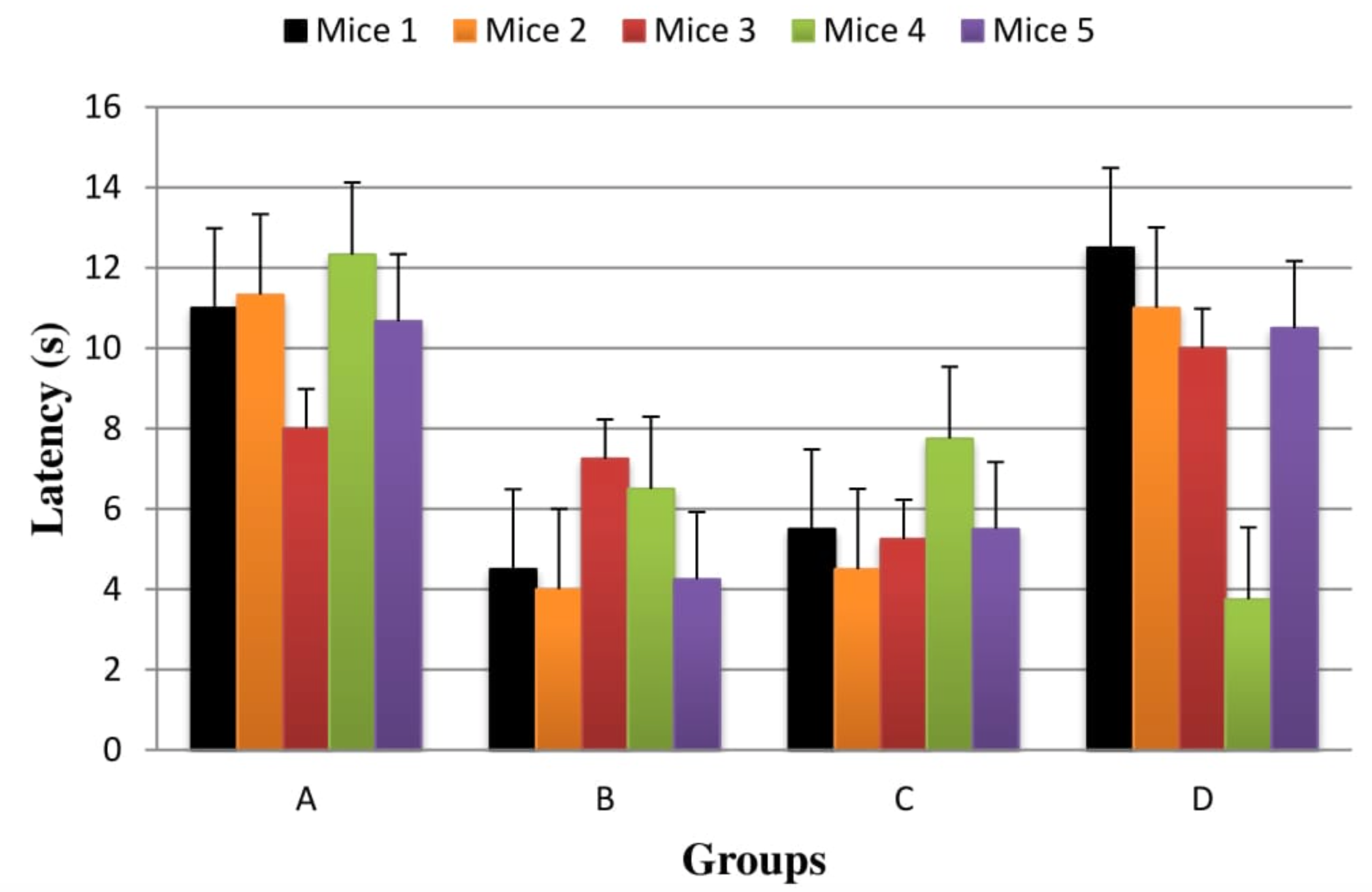
This test is used to measure the sensorimotor function of mice. PD motor tests provide a good read-out of neurological function. In case of Group D, it was seen that this group was statistically insignificant than Group B (P=0.409) while it was also noted that this group showed better neurological function as compared to Group C. However, results were also found insignificant for Group C as compared to Group B (P=1.00). Figure 3
Swim Test
The forced swim test is a rat Motor-function test utilized for assessment of stimulant medications, upper adequacy of new mixes, and trial controls that are gone for rendering or anticipating depressive-like states.
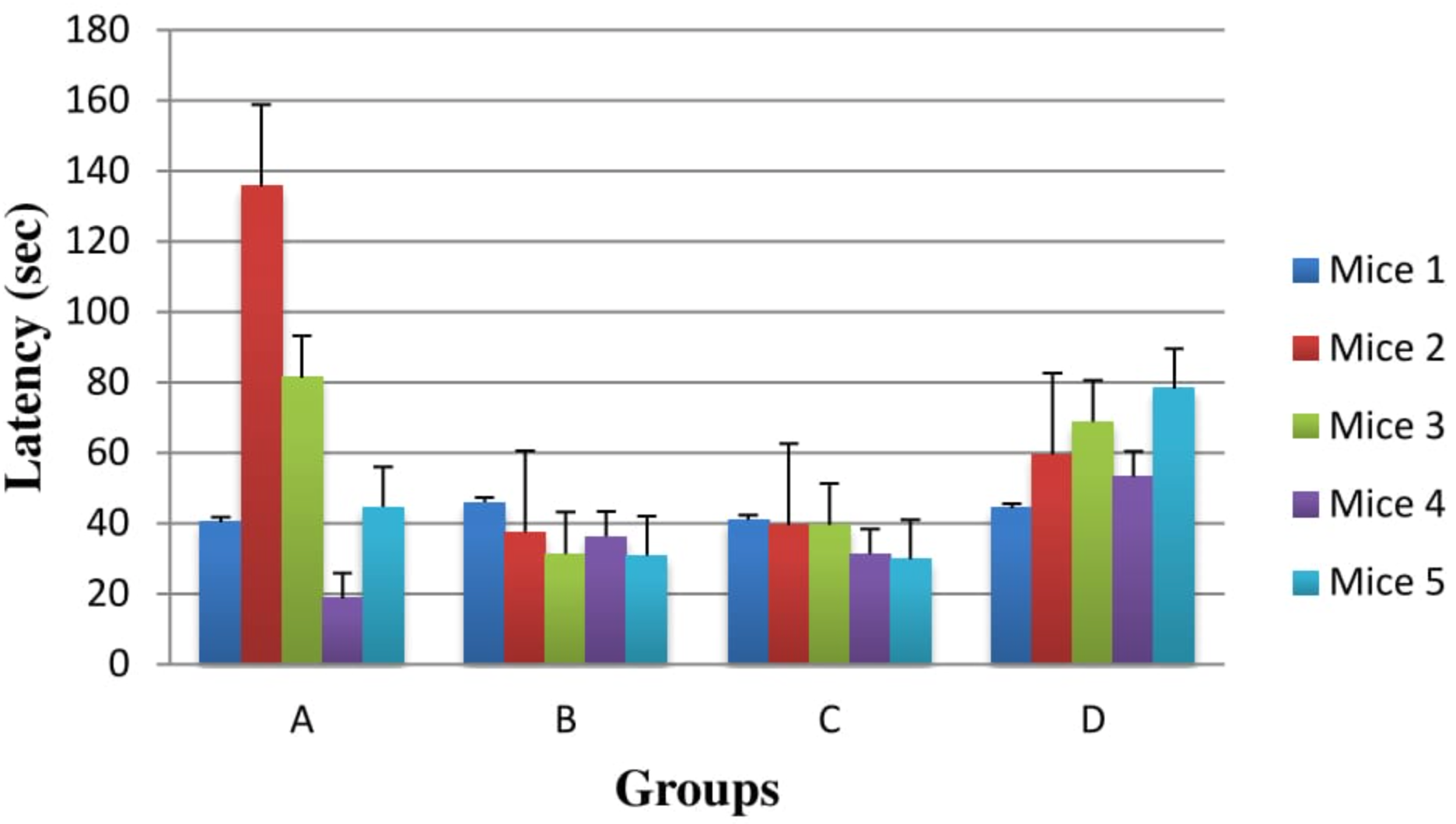
In case of Swim Test, Group D is statistically insignificant in association with Group C (P=0.284) and Group B (P=0.221). Therefore, it was observed that results were not statistically significant among all four groups in case of swim test as demonstrated in Figure 4 . Consequently in our study, it was observed that Pyridine-3-carboxylic acid 15 mg/kg/day PO dose is found effective as compared to Pyridine-3-carboxylic acid 7.15 mg/kg/day PO dose.
Discussion
The principle objective of exploration study displayed in this postulation is to describe the impacts of HP in the improvement of PD models and Motor-function undertakings some time recently, amid, and after treatment with Pyridine-3-carboxylic acid. In the present study, HP treatment altogether expanded vacuous biting development and tardive dyskinesia when contrasted with control mice. Neuroleptics act by blocking dopamine receptors Creese et al., 1976. Such blockage results in expanded dopamine turnover, which thus prompts expanded generation of hydrogen peroxide, bringing about oxidative anxiety Chauhan et al., 2004Elkashef and Wyatt, 1999. Existing proof shows that inordinate creation of free radicals is connected with interminable neuroleptic utilize and may add to the onset of tardive dyskinesia and other development issue, for example, dystonias and Parkinsonism Burger et al., 2005.
During motor-function modulation, an important parameter for treatment effectiveness is neuromuscular strength. Wire hanging test was performed to assess the neuromuscular ability of mice. It was observed during experimentation that Group D showed more neuromuscular strength than Group C as shown in Figure 1 . However, results of group D were not statistically much significant (P=0.082) in comparison with Group C. In one of the study where Effect of hypericum hookeranium on HP induced neuromuscular weakness was tested by wire hang test. Less latency to fall by releasing the wire soon indicates the apathetic state in the induced animal. The latency in falling represent the improved neuromuscular strength in 400 mg/kg EEHH treated animals with the same effect as that of the standard drug scopolamine. Before treatment animals had excellent neuromuscular activity, reduced by the treatment of Haloperidol. hypericum hookeranium at the dose 400mg/kg significantly Pongiya et al., 2014. The effect of low doses of Pyridine-3-carboxylic acid show more promising effects as compared to hypericum hookeranium 400mg/kg in this study.
Grip strength test is a test of muscle strength using all fore limbs. However, in our research study, Grip strength test showed that Group D mice had strong grip as compared to Group C and Group B mice as shown in Figure 2 . Results were found statistically significant for Group D in comparison with Group C (P=0.047). It was also observed during study that Group B had shown least strength of grip than Group A (P=0.005) as demonstrated in Figure 2 . The dose of 15mg/kg on HP induced mice model of PD show more promising effects during grip strength test as compared to 7.15 mg/kg. In one of the study, same tests were applied on mice model to check the impacts of HP on hold quality test are outlined at the lower left partition. There was a measurably huge general treatment impact and a pattern toward expanded grasp quality in the haloperidol-induced mice when subjected to motor integration tests. Grip strength test showed a decrease in muscle coordination which could be due to a loss of muscular strength.
Treatment with MECD showed a significant improvement in the muscle coordination as there is an increase in retention time and fall-off time in grip strength test respectively. Locomotor activity was also studied using actophotometer in which MECD improved the photocells count which was significantly less in HP treated group Pavan et al., 2015.
Vertical pole test is used to measure the sensorimotor function of mice. PD motor tests provide a good read-out of neurological function. The vertical pole test provides the information about the level of catalepsy generated by HP in mice model of PD and its effects on mice during and after the treatment on vertical pole test. In one of the study, HP created the prolongation of T-turn and TLA as a marker of bradykinesia in mice and the prolongation kept going no less than 7 hr after HP treatment. Intraperitoneal co-pretreatment with L-DOPA (400 mg/kg) + carbidopa (10 mg/kg) in mice diminished the catalepsy instigated by HP at a measurements of 0.125 mg/kg, while co-pretreatment with L-DOPA (200 and 400 mg/kg) + carbidopa (10 mg/kg) dosage conditionally diminished the HP (0.125 mg/kg) actuated bradykinesia. The impact of LDOPA + carbidopa in post-test was more purported than that in catalepsy test Kobayashi et al., 1997. The treatment with Pyridine-3-carboxylic acid in our research study show significant results in mice model of PD. In case of Group D, it was seen that this group was statistically insignificant than Group B (P=0.409) while it was also noted that this group showed better neurological function as compared to Group C. However, results were also found insignificant for Group C as compared to Group B (P=1.000). The data show that the both doses of Pyridine-3-carboxylic acid did not put much influence in this analytical parameter.
The forced swim test were utilized for assessment of upper medications, energizer viability of new mixes, and trial controls that are gone for rendering or counteracting depressive-like states. In one of the research study, swim test was used to evaluate the effectiveness of S-acetylcysteine on HP induced mice model of PD. In case of Swim Test, Group D is statistically insignificant in association with Group C (P=0.284) and Group B (P=0.221). Therefore, it was observed that results were not statistically significant among all four groups in case of swim test as demonstrated in figure 4. Consequently, in our study, it was observed that Pyridine-3-carboxylic acid15mg/kg/day PO dose is found effective as compared to Pyridine-3-carboxylic acid7.15mg/kg/day PO dose..
Conclusion
In the present study, the activities of mice get reduced after the administration of haloperidol due to its effect on dopamine blockade and development of symptoms of PD and Motor-function changes. The findings of the present study suggested that the use of Pyridine-3-carboxylic acid may be helpful as an adjunct therapy with standard therapy of Levodopa/Carbidopa in PD patients and can also decrease the level of association of free radicals in the improvement of neuroleptic prompted PD.
Abbreviations
EEHH: ethanolic extract of hypericum hookeranium
HP: haloperidol
I.P: intraperitoneal
L-DOPA: levo dopa
MPTP: 1-methyl-4-phenyl-1,2,3,6-tetrahydroxypyridine
NAD: nicotinamide adenine dinucleotide
PD: Parkinson's disease
PO: per oral
Author contribution
Atif Saeed, Arsalan Ali and Muhammad Yousaf performed lab work and behavioral testing. Awais Ali Zaidi give the concept, designed experiment and supervised the project. Lubna Shakir helped in data acquisition and experiment conditions optimization Mahtab Ahmad Khan analyzed data and gave final approval of the project
References
-
A.
Aartsma-Rus,
M.
van Putten.
Assessing functional performance in the mdx mouse model. JoVE (Journal of Visualized Experiments).
2014;
:
e51303-e51303
.
-
H.
Bernheimer,
W.
Birkmayer,
O.
Hornykiewicz,
K.
Jellinger,
F.
Seitelberger.
Brain dopamine and the syndromes of Parkinson and Huntington Clinical, morphological and neurochemical correlations. Journal of the neurological sciences.
1973;
20
:
415-455
.
-
M.E.
Burger,
R.
Fachinetto,
G.
Zeni,
J.B.
Rocha.
Ebselen attenuates haloperidol-induced orofacial dyskinesia and oxidative stress in rat brain. Pharmacology Biochemistry and Behavior.
2005;
81
:
608-615
.
-
A.
Chauhan,
V.
Chauhan,
W.T.
Brown,
I.
Cohen.
Oxidative stress in autism: Increased lipid peroxidation and reduced serum levels of ceruloplasmin and transferrin-the antioxidant proteins. Life sciences.
2004;
75
:
2539-2549
.
-
I.
Creese,
D.R.
Burt,
S.H.
Snyder.
Dopamine receptor binding predicts clinical and pharmacological potencies of antischizophrenic drugs. science.
1976;
192
:
481-483
.
-
A.M.
Elkashef,
R.J.
Wyatt.
Tardive dyskinesia: possible involvement of free radicals and treatment with vitamin E. Schizophrenia bulletin.
1999;
25
:
731-740
.
-
P.O.
Fernagut,
E.
Diguet,
B.
Labattu,
F.
Tison.
A simple method to measure stride length as an index of nigrostriatal dysfunction in mice. Journal of neuroscience methods.
2002;
113
:
123-130
.
-
A.
Gurakar,
J.M.
Hoeg,
G.
Kostner,
N.M.
Papadopoulos,
H.B.
Brewer.
Levels of lipoprotein Lp (a) decline with neomycin and niacin treatment. Atherosclerosis.
1985;
57
:
293-301
.
-
D.
Hirtz,
D.
Thurman,
K.
Gwinn-Hardy,
M.
Mohamed,
A.
Chaudhuri,
R.
Zalutsky.
How common are the “common” neurologic disorders?. Neurology.
2007;
68
:
326-337
.
-
P.A.
Kempster,
B.
Hurwitz,
A.J.
Lees.
A new look at James Parkinson's Essay on the Shaking Palsy. Neurology.
2007;
69
:
482-485
.
-
T.J.
Klein,
M.A.
Lewis.
A physical model of sensorimotor interactions during locomotion. Journal of neural engineering.
2012;
9
:
046011
.
-
T.
Klockgether.
Parkinson’s disease: clinical aspects. Cell and tissue research.
2004;
318
:
115-120
.
-
T.
Kobayashi,
T.
Araki,
Y.
Itoyama,
M.
Takeshita,
T.
Ohta,
Y.
Oshima.
Effects of L-dopa and bromocriptine on haloperidol-induced motor deficits in mice. Life sciences.
1997;
61
:
2529-2538
.
-
S.
Manikandaselvi,
R.
Mahalakshmi,
R.
Thinagarbabu,
A.
Angumeenal.
Neuroprotective activity of S-Allylcysteine on Haloperidol induced Parkinson’s disease in albino mice. Int J Pharm Technol Res.
2012;
4
:
669-675
.
-
J.
Molina,
F.
Jiménez-Jiménez,
J.
Navarro,
C.
Vargas,
P.
Gomez,
J.
Benito-León,
M.
Ortí-Pareja,
E.
Cisneros,
J.
Arenas.
Cerebrospinal fluid nitrate levels in patients with Parkinson's disease. Acta neurologica scandinavica.
1996;
93
:
123-126
.
-
M.C.
Morris,
D.A.
Evans,
J.L.
Bienias,
P.A.
Scherr,
C.C.
Tangney,
L.E.
Hebert,
D.
Bennett,
R.S.
Wilson,
N.
Aggarwal.
Dietary niacin and the risk of incident Alzheimer’s disease and of cognitive decline. Journal of.
2004;
Neurology
:
Neurosurgery & Psychiatry 75, 1093-1099
.
-
P.S.
Naidu,
A.
Singh,
S.K.
Kulkarni.
Quercetin, a bioflavonoid, attenuates haloperidol-induced orofacial dyskinesia. Neuropharmacology.
2003;
44
:
1100-1106
.
-
T.
Pavan,
K.
Manasa,
T.
Tamilanban,
V.
Alagarsamy.
Effect of Methanolic extract of Canscora decussata on Haloperidol-Induced Motor deficits in Albino mice. Int J Pharm Sci Rev Res.
2015;
35
:
7-11
.
-
U.D.
Pongiya,
B.M.
Kandanath,
Y.R.
Rao.
Efficacy of hypericum hookerianum in reversing the symptoms of haloperidol induced tardive dyskenesia, catatonia and catalepsy in swiss albino mice-behavioural analysis report. World Journal of Pharmacy And Pharmaceutical Sciences.
2014;
3
:
1682-1705
.
-
M.
Rahman,
S.
Muhammad,
M.A.
Khan,
H.
Chen,
D.A.
Ridder,
H.
Muller-Fielitz,
B.
Pokorna,
T.
Vollbrandt,
I.
Stolting,
R.
Nadrowitz.
The beta-hydroxybutyrate receptor HCA2 activates a neuroprotective subset of macrophages. Nature communications.
2014;
5
:
3944
.
-
B.
Scholtissen,
F.
Verhey,
H.
Steinbusch,
A.
Leentjens.
Serotonergic mechanisms in Parkinson’s disease: opposing results from preclinical and clinical data. Journal of neural transmission.
2006;
113
:
59-73
.
-
H.
Sheidaei.
Buspirone improves haloperidol-induced Parkinson disease in mice through 5-HT1A recaptors. DARU: Journal of Faculty of.
2010;
Pharmacy
:
Tehran University of Medical Sciences 18, 41
.
-
S.
Wang,
L.-f.
Hu,
Y.
Yang,
J.-h.
Ding,
G.
Hu.
Studies of ATP-sensitive potassium channels on 6-hydroxydopamine and haloperidol rat models of Parkinson's disease: implications for treating Parkinson's disease?. Neuropharmacology.
2005;
48
:
984-992
.
-
A.A.
Zaidi,
T.A.
Khan,
L.
Shakir,
M.A.
Khan,
M.
Yousaf,
A.
Ali.
Evaluation of C. cassia Effectiveness in Behavioral Modulation of Haloperidol Induced Parkinson’s Disease (Mice Model). British Journal of Pharmaceutical Research.
2016a;
6
:
1-7
.
-
A.A.
Zaidi,
L.
Shakir,
T.A.
Khan,
M.A.
Khan,
A.
Ali,
A.U.
Rehman.
Haloperidol leads to torse de pointes in schizophrenic pool. European Journal of Pharmaceutica And Medical Research.
2016b;
3
:
84-91
.
Comments

Downloads
Article Details
Volume & Issue : Vol 4 No 05 (2017)
Page No.: 1305-1317
Published on: 2017-05-22
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 9118 times
- Download PDF downloaded - 1936 times
- View Article downloaded - 26 times
 Biomedpress
Biomedpress