Abstract
Introduction: During the past two decades, eradication rates with triple therapy for Helicobacter (H.) pylori eradication have decreased. The decline of effectiveness of the triple therapy have led to investigations to achieve more effective and safe therapeutic strategies. Sequential, concomitant and hybrid therapeutic regimens are new therapies that have been introduced over the past two decades. The purpose of this study was to compare levofloxacin-based sequential therapy versus classic triple therapy in H. pylori eradication in a randomized clinical trial.
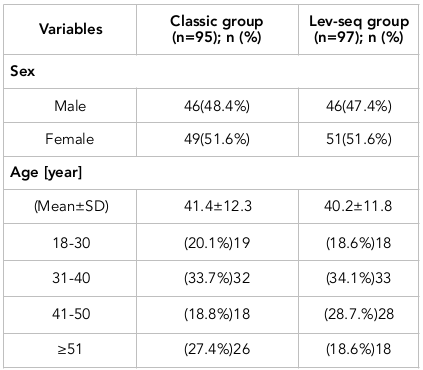
Methods: All eligible cases were divided into two groups using a randomized block method. The first group (classic group) was treated with triple therapy; patients received omeprazole, amoxicillin and clarithromycin for 14 days. The second group received the levofloxacin-based sequential (lev-seq) regimen; the patients were treated with omeprazole and amoxicillin for the first 7 days and then with omeprazole, levofloxacin and metronidazole for the latter 7 days. In this clinical trial, 200 patients with H. pylori infection were enrolled. Ultimately, 192 patients completed the study. Of these, 95 patients (46 males and 49 females) were treated with triple therapy (classic group) and 97 patients (46 males and 51 females) were treated with the lev-seq regimen. The average age in the classic group and lev-seq groups were 41.4±12.4 years and 40.2±11.8 years, respectively.
Results: The levofloxacin based sequential regimen was more effective than triple therapy regimen. It is recommended that the lev-seq regimen be used as the first-line therapy, especially in areas with high levels of resistance to clarithromycin (over 20%).
Conclusion: Further studies with large numbers of samples are required to be conducted in different parts of the country for further evaluation of efficacy of this regimen.
Introduction
During the past two decades, triple therapy for Helicobacter (H.) pylori have yielded low eradication rates Chey and Wong, 2007Park et al., 2016. The decline of the effectiveness of triple therapy has heightened the need to investigate more ideal, effective and safe therapeutic regimens. Sequential, concomitant, and hybrid therapeutic regimens are a few of the new therapies that have been introduced over the past two decades Malfertheiner et al., 2017Park et al., 2016. A sequential regimen involves a 2-step drug regimen. For instance, over the first 5 days PPI and amoxicillin are administered, and in the latter 5 days PPI, clarithromycin, and metronidazole (imidazole) are administered De Francesco et al., 2010Zullo et al., 2000. Preliminary studies have demonstrated that the eradication rate of sequential regimen is very high (over 95%) Vaira et al., 2007Vakil and Vaira, 2008Zullo et al., 2000. However, even recently, studies continue to suggest that eradication with classic sequential regimen is not entirely effective Molina-Infante et al., 2010Romano et al., 2010. Thus, in this study, we took a different approach and evaluated a levofloxacin-based sequential regimen [modified sequential]; in the second (latter) step of the sequential drug regimen, levofloxacin was administered instead of clarithromycin Molina-Infante et al., 2010. The resistance of H. pylori strains to levofloxacin is very low (AzadBakht, 2003). Indeed, in Iran the level of resistance to levofloxacin has also been reported to be low (3.5%) Khademi et al., 2015. It appears that the levofloxacin-based sequential regimen can be a viable alternative in the treatment of H. pylori infections, especially in areas with high levels of clarithromycin resistance. The aim of this study was to compare the eradication rate of levofloxacin-based sequential regimes and classic triple therapy.
Materials-Methods
This clinical trial was conducted from October 2015 to March 2016 at Shohadaye Ashayer Hospital in Khorramabad (West Iran). The study population includes dyspeptic patients referred to the gastrointestinal clinic.
Patients over 18 years with histologically proven H. pylori infections were included in the study. Patients who had been treated previously for H. Pylori infection and those with a history of sensitivity to antibiotics were excluded. Additionally, people with a history of gastrectomy and those with severe underlying diseases, such as renal failure and liver cirrhosis, were excluded, as were pregnant and lactating women. The written consents were received from all patients who were eligible to participate in the study. All eligible cases were divided into two groups using a randomized block method. The first group (classic) was treated with triple therapy; these patients received omeprazole, amoxicillin, and clarithromycin for 14 days. The second group (lev-seq) was treated with levofloxacin-based sequential regimen; these patients received omeprazole and amoxicillin during the first 7 days, and omeprazole, levofloxacin and metronidazole for the latter 7 days.
Patients in both groups received an educational fact sheet on the use of drugs and their possible side effects. The patients were advised to visit their doctor once during the treatment period and also at the end of the treatment period. All patients were visited by a doctor twice. After the history and physical examinations by the doctor, patients were monitored for side effects and tolerance. Besides information obtained from history and physical examinations, patient adherence to the study and side effects to the drugs were recorded. Drug side effects were reported by the patients in a self-reporting questionnaire. The adherence rate was evaluated with questions about the number of consumed pills. If the patient consumed more than 90% of the prescription drugs, then adherence was considered acceptable. For all patients, the urea breath test (Urea Breath Test) was performed 6 weeks after completion of treatment. The success of the therapeutic regimen indicates that the cases were negative; UBT. This study has been recorded in the IRCT number 2015082323736N1.
Statistical analysis
The results of this study were analyzed in an intention-to-treat population and a per-protocol population. SPSS version 21 (IBM Inc.; New York, USA) was used for the statistical analyses, in which the eradication rate was analyzed by chi-square test and Student’s t-test, and the factors affecting eradication were analyzed by multivariable logistic regression. Any P-value of <0.05 was deemed as statistically significant.
Results
In this clinical trial, 200 patients with H. pylori infection were enrolled. Among these, 4 patients left the study due to severe side effects and 4 others did not return for follow-up. Ultimately, 192 patients completed the study. Of these, 95 patients (46 males and 49 females) were treated with triple therapy (classic group) while 97 patients (46 males and 51 females) were treated with the levofloxacin-based sequential regimen (lev-seq). The average age of the classic group and lev-seq groups were 41.4±12.4 years and 40.2±11.8 years, respectively. The demographic characteristics of the patients participating in the study are presented in Table 1 .
Primary outcome (H. Pylori eradication)
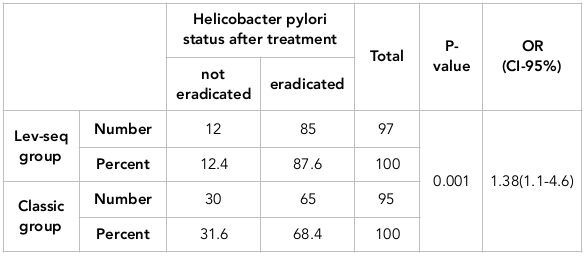
The effects of the two groups (classic and lev-seq) as H. pylori therapy are presented in Table 2 . The eradication rate in the classic group was calculated as 68.4% (using per protocol analysis) and 65% (using the intention-to-treat analysis). In contrast, the eradication rate of the lev-seq regimen was 87.6% (using per protocol analysis) and 85% (using the intention-to-treat analysis). The eradication rate of H. pylori in the Lev-seq group was higher than that of the classic group; this difference was statistically significant (p=0.001). Moreover, the H. pylori eradication probability with the lev-seq regimen was 1.38-fold greater than that of the triple therapy regimen (OR=1.38 (1.1-4.6)).
Adherence and adverse events
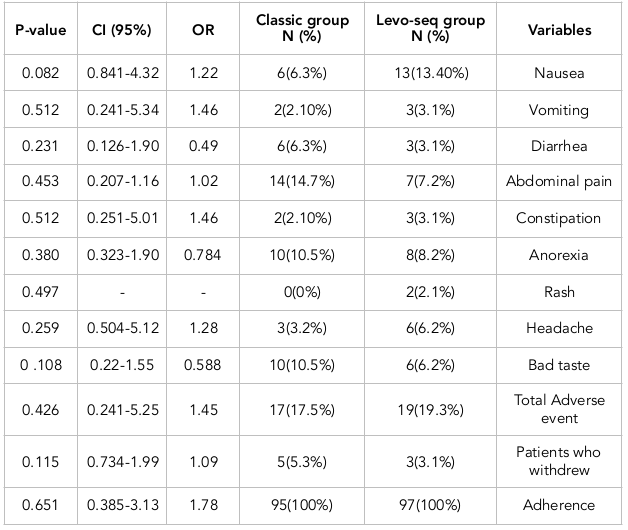
The frequency of complications occurring with either treatment regimens are presented in Table 3 . Of note, 17 (17.8%) patients of the classic group and 19 (19.5%) patients of the lev-seq group experienced drug complications. There was no statistically significant difference between the groups (P =0.067).
The incidence of adverse events were not significantly different between the classic and lev-seq groups. The most common drug complications reported in the classic group included abdominal pain, loss of appetite and a bad oral taste, while the most common complications of the lev-seq group included nausea, anorexia and abdominal pain.
The adherence rate to the classic regimen and Lev-seq regimen were 95% and 97%, respectively. The adherence rates in both groups were acceptable although the difference between the two groups was not significant (P>0.05%).
Factors affecting eradication rate
Using logistic regression to eliminate confounding factors, the factors that may have an impact on treatment success rates were examined. Based on the results, multivariant, age, sex, education, smoking, and alcohol had no effects on the eradication of Helicobacter.
Discussion
This clinical trial, conducted on 192 patients, aimed to compare the efficacy of levofloxacin-based sequential and triple therapy regimens. Based on the results of this study, the lev-seq regimen is more effective than triple therapy (p= 0.001). H. pylori eradication rates for the lev-seq regimen were 87.6% and 85%, respectively, by PP and ITT analysis. The eradication rates of triple therapy with PP and ITT analysis were 68.4% and 65%, respectively. Our study suggests that eradication rates with triple therapy are suboptimal (less than 80%). The results of our study are similar with the results of other studies Gatta et al., 2013Jafri et al., 2008Moradniani et al., 2017b.
In recent years, the eradication rates using triple therapy has significantly decreased Chey and Wong, 2007Malfertheiner et al., 2017Moradniani et al., 2017b. Most studies in Asia have also demonstrated the reduction of success rate with triple therapy Fakheri et al., 2014Moradniani et al., 2017aMoradniani et al., 2017b. The main reason for reducing the effectiveness of triple therapy is the increasing resistance to antibiotics, particularly clarithromycin De Francesco et al., 2010Jafri et al., 2008Park et al., 2016. In Iran, like other countries, the rate of resistance to clarithromycin is increasing. Resistance to clarithromycin has reached from 4.6% in 2000 to 22.4% in 2013. Azadbakht S, 2017Khademi et al., 2015. Our study shows that the lev-seq regimen is much more effective than the triple therapy regimen. The results of our study are quite consistent with other studies that have been done in other country Kale-Pradhan et al., 2015Molina-Infante et al., 2010Ozdil et al., 2011Romano et al., 2010.
A recent meta-analysis reported the eradication rate of lev-seq regimen as 87.8% and the eradication rate using triple therapy as 71% Gatta et al., 2013. A clinical trial in Spain has reported the eradication rate using the Lev-seq regime as 82.6% and eradication rate using triple therapy as 64% Lee et al., 2015. As well, another study in Turkey has demonstrated the eradication rates using the lev-seq regimen and triple therapy as 72.5% and 92%, respectively Polat et al., 2012. The main cause of the high rates of eradication using the lev-seq regime is the low rate of resistance to levofloxacin De Francesco et al., 2010Park et al., 2016. In a recent study in Iran, it was reported that resistance to levofloxacin was 3.5% Khademi et al., 2015. Levofloxacin is a fluoroquinolone drug which influences H. pylori strains that are resistant to clarithromycin Kale-Pradhan et al., 2015. Sequential regimens based on levofloxacin is a viable option and can be an ideal alternative for the treatment of H. pylori infection, especially in areas where resistance to clarithromycin is high Kale-Pradhan et al., 2015Malfertheiner et al., 2017.
The results of our study show that the rate of complications in levofloxacin-based sequential regimen and triple therapy wasn’t significantly different. The most common side effects were reported in triple therapy to include abdominal pain, loss of appetite and bad oral taste. The most common side effects of the lev-seq regimen include anorexia, nausea and abdominal pain. Our results are consistent with most studies Moghadaszadeh, 2012Molina-Infante et al., 2010Moradniani et al., 2017bRomano et al., 2010Seyyedmajidi et al., 2011. In such studies, no significant difference was found between the two groups in terms of side effects Moghadaszadeh, 2012Molina-Infante et al., 2010Moradniani et al., 2017bRomano et al., 2010.
The high number of consumed drugs, particularly the use of different antibiotics, can cause various side effects. In a study that was conducted by Eisig and colleagues in Brazil, diarrhea, abdominal pain and nausea were expressed, along with other common side effects Eisig et al., 2011. Roman and colleagues in Italy showed that there is no statistically significant difference between lev-seq and triple therapy regimens in terms of side effects Romano et al., 2010.
Our results show that the eradication rate of H. pylori infection has no association with age, sex, smoking, alcohol consumption, education level and side effects. These factors have no impact on the eradication of H. pylori. Results of our study are consistent with the results of other studies Kale-Pradhan et al., 2015Moradniani et al., 2017aMoradniani et al., 2017bPolat et al., 2012Romano et al., 2010. However some studies have shown that age and cigarette smoking may have an impact on the eradication rate of H. pylori infection Eisig et al., 2011Molina-Infante et al., 2010.
Levofloxacin-based sequential regimen is more effective than triple therapy regimen. It is recommended that lev-seq regimen be used as the first-line therapy, especially in areas with high levels of resistance to clarithromycin (over 20%). More studies with larger numbers of samples are needed to further evaluate the efficacy of this regimen in different parts of the country.
Conclusion
Levofloxacin-based sequential regimen is more effective than triple therapy regimen. It is recommended that lev-seq regimen be used as the first-line therapy, especially in areas with high levels of resistance to clarithromycin (over 20%). More studies with larger numbers of samples are needed to further evaluate the efficacy of this regimen in different parts of the country.
Abbreviations
H. pylori : Helicobacter pylori
Leva seq : Levofloxacin-based sequential
Author Contribution
All authors contributed to the design of the research. MSH, SH SH, SA, MM, HM, SJ, MMR, ZMS write, edited and revised it and PB analysis it. All authors reviewed and commented on final draft.
References
-
W. D.
Chey,
B. C.
Wong.
American College of Gastroenterology guideline on the management of Helicobacter pylori infection. The American Journal of Gastroenterology.
2007;
102(8)
:
1808-1825
.
View Article Google Scholar -
J. Y.
Park,
K. B.
Dunbar,
M.
Mitui,
C. A.
Arnold,
D. M.
Lam-Himlin,
M. A.
Valasek,
C. D.
Doern.
Helicobacter pylori Clarithromycin resistance and treatment failure are common in the USA. Digestive Diseases and Sciences.
2016;
61(8)
:
2373-2380
.
View Article Google Scholar -
P.
Malfertheiner,
F.
Megraud,
C. A.
O’Morain.
European Helicobacter and Microbiota Study Group and Consensus panel. Management of Helicobacter pylori infection-the Maastricht V/Florence Consensus Report. Gut.
2017;
66
:
6-30
.
-
V.
De Francesco,
F.
Giorgio,
C.
Hassan.
Worldwide H. pylori antibiotic resistance: A systematic review. Journal of Gastrointestinal and Liver Diseases; JGLD.
2010;
19
:
409-414
.
-
A.
Zullo,
V.
Rinaldi,
S.
Winn,
Meddi.,
Lionetti.,
Hassan.,
Attili..
A new highly effective short-term Therapy schedule for Helicobacter pylori eradication. Alimentary Pharmacology & Therapeutics.
2000;
14(6)
:
715-718
.
View Article Google Scholar -
N.
Vakil,
D.
Vaira.
Sequential therapy for Helicobacter pylori: Time to Consider making the switch?. Journal of the American Medical Association.
2008;
300(11)
:
1346-1347
.
-
D.
Vaira,
A.
Zullo,
N.
Vakil,
L.
Gatta,
C.
Ricci,
F.
Perna,
S.
Morini.
Sequential therapy versus standard Triple-drug therapy for Helicobacter pylori eradication: A randomized Trial. Annals of Internal Medicine.
2007;
146(8)
:
556-563
.
View Article Google Scholar -
J.
Molina-Infante,
B.
Perez-Gallardo,
M.
Fernandez-Bermejo.
Clinical trial: Clarithromycin vs. levofloxacin in first-line triple and sequential regimens for Helicobacter pylori eradication. Alimentary Pharmacology & Therapeutics.
2010;
31
:
1077-1084
.
-
M.
Romano,
A.
Cuomo,
A. G.
Gravina,
A.
Miranda,
M. R.
Lovene,
A.
Tiso.
Empirical Levofloxacin-containing versus Clarithromycin-containing sequential therapy for Helicobacter pylori eradication: A randomised trial. Gut.
2010;
59(11)
:
1465-1470
.
View Article Google Scholar -
S
Azadbakht,
|M
Moradniani,
Z
Mirbeik Sabzevari,
M
Hassani,
M J.
Tarrahi.
The Prevalence of Intestinal Metaplasia in Dyspeptic Patients. [IJBR]. International Journal of Advanced Biotechnology and Research.
2017;
8(3)
:
2156-2165
.
-
F.
Khademi,
F.
Poursina,
E.
Hosseini,
M.
Akbari,
H.
Ghasemian Safaei.
Helicobacter pylori in Iran: A systematic review on the antibiotic resistance. Iranian Journal of Basic Medical Sciences.
2015;
18
:
2-7
.
-
N. S.
Jafri,
C. A.
Hornung,
C. W.
Howden.
Meta-analysis: Sequential therapy appears superior to standard therapy for Helicobacter pylori infection in patients naive to Treatment. Annals of Internal Medicine.
2008;
148(12)
:
923-931
.
View Article Google Scholar -
L.
Gatta,
N.
Vakil,
D.
Vaira,
C.
Scarpignato.
Global eradication rates for Helicobacter pylori infection: Systematic review and meta-analysis of sequential therapy. BMJ (Clinical Research Ed.).
2013;
347(aug07 1)
:
f4587
.
-
M.
Moradniani,
Z.
Mirbeik-Sabzevari,
M.
Bahmani,
S.
Azadbakht,
S.
Jaferian.
Comparison of 7 - day concomitant therapy regimen versus classic triple therapy regimen in Helicobacter pylori eradication: A randomized clinical trial. Int J Pharm Sci Res.
2017;
8(8)
:
3568-3573
.
-
H.
Fakheri,
Z.
Bari,
M.
Aarabi,
R.
Malekzadeh.
Helicobacter pylori eradication in West Asia: A review. World Journal of Gastroenterology.
2014;
20(30)
:
10355-10367
.
View Article Google Scholar -
M.
Moradniani,
S.
Azadbakht,
S.
Jaferian.
levofloxacin versus clarithromycin sequential therapy. Gastroenterology and Hepatology from Bed To Bench.
2017;
:
10
.
-
K.
Ozdil,
T.
Calhan,
A.
Sahin,
E.
Şenateş,
R.
Kahraman,
B.
Yüzbaşıoğlu,
H. M.
Sökmen.
Levofloxacin based sequential and triple therapy compared with standard plus probiotic combination for Helicobacter pylori eradication. Hepato-Gastroenterology.
2011;
58(109)
:
1148-1152
.
View Article Google Scholar -
P. B.
Kale-Pradhan,
A.
Mihaescu,
S. M.
Wilhelm.
Fluoroquinolone sequential Therapy for helicobacter pylori: A meta-analysis. Pharmacotherapy.
2015;
35(8)
:
719-730
.
View Article Google Scholar -
H.
Lee,
S. N.
Hong,
B. H.
Min,
J. H.
Lee,
P. L.
Rhee,
Y. C.
Lee,
J. J.
Kim.
Comparison of efficacy and safety of Levofloxacin-containing versus standard sequential therapy in eradication of Helicobacter pylori infection in Korea. Digestive and Liver Disease.
2015;
47(2)
:
114-118
.
View Article Google Scholar -
Z.
Polat,
A.
Kadayifci,
M.
Kantarcioglu,
A.
Ozcan,
O.
Emer,
A.
Uygun.
Comparison of levofloxacin-containing sequential and standard triple therapies for the eradication of Helicobacter pylori. European Journal of Internal Medicine.
2012;
23(2)
:
165-168
.
View Article Google Scholar -
E.
Moghadaszadeh MFattahi,
MH
Somi,
M
Khaoshbaten,
T
Rosta.
The comparison between two treatment methods for H. pylori eradication with Two-week sequential regimens. J Arak Univer Med Sci..
2012;
15(61)
:
93-99
.
-
M.
Seyyedmajidi,
K.
Falaknazi,
D.
Mirsattari,
H.
Zojaji,
M.
Roshani,
F.
Lahmi,
M.
Zali.
Correlation2 between creatinine clearance and Helicobacter pylori infection eradication with sequential and triple therapeutic regimens: A randomised clinical trial. Arab Journal of Gastroenterology.
2011;
12(3)
:
150-153
.
View Article Google Scholar -
J. N.
Eisig,
F. M.
Silva,
R. C.
Barbuti,
T.
Navarro-Rodriguez,
J. P.
Moraes-Filho,
J. Jr.
Pedrazzoli.
Helicobacter pylori antibiotic resistance in Brazil: Clarithromycin is still a good option. Arquivos de Gastroenterologia.
2011;
48(4)
:
261-264
.
View Article Google Scholar -
M.
Moradniani,
Z.
Mirbeik-Sabzevari,
M.
Bahmani,
S.
Azadbakht,
S.
Jaferian.
Comparison of 7 - day concomitant therapy regimen versus classic triple therapy regimen in Helicobacter pylori eradication: A randomized clinical trial. Int J Pharm Sci Res.
2017;
8(8)
:
3568-3573
.
-
M.
Moradniani,
S.
Azadbakht,
S.
Jaferian.
levofloxacin versus clarithromycin sequential therapy. Gastroenterology and Hepatology from Bed To Bench.
2017;
:
10
.
Comments
Downloads
Article Details
Volume & Issue : Vol 4 No 11 (2017)
Page No.: 1785-1794
Published on: 2017-11-30
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
- HTML viewed - 5108 times
- Download PDF downloaded - 1369 times
- View Article downloaded - 10 times
 Biomedpress
Biomedpress