Abstract
Background: The potential use of plasma galectin-3 level as a of myocardial changes in the development of hypertrophy in males with essential hypertension (EH), carriers of polymorphic variants of II type 1 receptor gene, was studied. The level of brain peptide (BNP), as well as the parameters of central and systemic, and structural and functional parameters of the heart in carriers of polymorphic variants of II type 1 receptor gene, were studied as reference.
Methods: The study included 79 individuals with no signs of cardiovascular disease, and 62 males with stage II EH, aged 40-60, who were residents of Ukraine. All patients were treated at the Military Medical Clinical Center of Central Region of Air Forces of Ukraine and Vinnytsia Regional Specialized Clinical Hospital of Radiation Protection of the Ministry of Health of Ukraine, from December 2013 to June 2015. The patients were studied during their treatment at the in-patient department. Anesthetic, clinical and laboratory findings, and measures were entered into a special chart. Biochemical analyses were done upon admission of the patients to the hospital. Immunologic, and instrumental methods of examination were used as well. Statistical analyses of the results obtained were done on a personal computer using standard statistical package STATISTICA 10. Microsoft Excel was used for primary preparations of tables and intermediate calculations.
Results: In patients with EH and myocardial hypertrophy, the C allele of angiotensin II type 1 receptor gene was found to be dominating. They had higher levels of both galectin-3 and BNP. The threshold levels of galectin-3 and BNP for screening diagnosis of EH with myocardial hypertrophy in males (who were residents of the Podillya region of Ukraine) were evaluated.
Background
High blood pressure (BP) is one of the main determinants of cardiovascular risk according to the European Society of Cardiology (ESC) 2016 1. Despite the proven efficacy of anti-hypertensive therapy and ease of diagnosis and monitoring, a significant proportion of patients with essential hypertension (EH) remains undetected or is treated inadequately, according to the American Society of Hypertension (ASH) in 2017 2.
EH is a multifactorial disease in which, theoretically, genetic factors play a leading role in its development; thus, genetic factors can be used in determining the level of cardiovascular risk 34. According to current statistical data, EH is the leading cardiovascular pathology in the Ukraine and worldwide.
According to the literature, in the vast majority of patients there is no single gene responsible for the onset of EH. Instead, 107 gene loci have been identified which are likely responsible for elevation of blood pressure (BP) 2. A number of gene single nucleotide polymorphisms (SNPs) are considered to be factors of hereditary predisposition to EH. Polymorphism of angiotensin II type 1 receptor (AT1R) gene is one of the most frequently studied phenomenon as it contributes to the hemodynamic effects of pressure in the renin-angiotensin-aldosterone system (RAAS) 56.
The RAAS activity, to a large extent, depends on the state of counteracting systems, such as the system of natriuretic peptides (NUP), particularly brain natriuretic peptide (BNP) 7. Nowadays, it is used as a biomarker that is helpful in distinguishing dyspnea of cardiac origin from that developed due to respiratory system pathology 1. Absolute blood concentration of the BNP peptide in patients with severe heart failure has been found to reflect the degree of individual cardiovascular risk 1. However, BNP has also been known to reflect the state of myocardium, and fibrosis of cardiomyocytes is one of the links of left ventricular hypertrophy (LVH).
Galectin-3, a member of galectin protein family, is also a potential biomarker for fibrosis 8. Galectin-3 concentration in blood was found to be maximal at the peak of myocardial fibrosis and inflammation 910. One of the limitations in assessing the prognostic value of galectin-3, as with BNP, is its dependence on sex- and age-related differences, as well as on renal glomerular filtration rate 1112.
The impact of pathogenetic hereditary factors (e.g. polymorphism of AT1R gene) on galectin-3 concentration requires further studies. Polymorphism of these receptors can become a "conditional trigger" for regulating the level of dependent biological substances that can serve as biomarkers of the above-mentioned processes and can be used in predicting cardiovascular risk. Indeed, a parallel study of can be helpful in clarifying the efficacy of galectin-3 as a potential biomarker of myocardial hyperthrophy in EH 131415. While changes of blood plasma BNP concentration in the development of LVH and carriage of polymorphic variants of AT1R gene have been shown previously in a number of published studies 16176, no studies have been conducted concerning the role of plasma galectin-3 concentration.
This study aimed to study the potential use of blood plasma galectin-3 concentration in male patients with EH (aged 40-60 years and carriers of polymorph variants of the AT1R gene) as a biomarker of myocardial hypertrophy.
Methods
Patients
The subjects evaluated in this study consisted of 79 men, aged 40-60 (average age of 57.06 ± 0.50 years) with no signs of cardiovascular disease, and 62 men (average age of 49.19 ± 0.66 years) with stage II hypertensive disease and/or myocardial hypertrophy but no clinical signs of chronic heart failure (CHF). The males of the control group and those with EH were represented by age. All of them were inpatients at Vinnytsia Regional Specialized Clinical Hospital of Radiation Protection of the Ministry of Health of Ukraine at the Military Medical Clinical Center of Central Region of the Air Forces of Ukraine; they were observed on an outpatient basis from December 2013 to June 2015.
The study entrance criteria were
Exclusion criteria were
The following research methods were used in the study
1. General clinical methods — assessment of the eligible individual’s health status.
2. Blood chemistry — determination of blood lipids, levels of blood urea, creatinine, glucose and electrolytes, and protein spectrum.
3. Immunologic methods — determination of galectin-3 and BNP levels in blood plasma by enzyme immunoassay.
4. Cytogenetic method — detection of polymorph variants of AT1R gene by polymerase chain reaction (PCR).
5. Instrumental methods of diagnosis — electrocardiography, and M-mode, B-mode and Doppler echocardiography to determine the structural and functional parameters of myocardium, cardiac stress test, and measurement of office BP.
6. Mathematical methods for statistical processing of the results obtained.
Ethical principles
All patients provided their written informed consent for participation in the study.
PCR assay for determination of the genotype of angiotensin II type 1 receptor ( AT1R) gene
To determine the alleles of the polymorphic site (A1166C) of the AT1R gene, genomic DNA was isolated from venous blood leukocytes using “Set for separation of DNA/RNA from blood serum or blood plasma” (Private enterprise “LitTech”, Russia).
Blood collection was done in fasting state from the cubital vein; 4 ml of whole blood was collected into cooled polypropylene tubes containing EDTA (1 mg/ml of blood). The collected blood was preserved at -20°С for no more than 6 months before analysis.
Genotyping of the polymorphic site (A1166C) of the AT1R gene was carried out by means of amplification, using the PCR method on amplificator “Tertsik” (“DNA-Technology” LLC, Russia). Two reactions of amplification with two pairs of allele specific oligonucleotide primers were carried out simultaneously: АТ1R-L (5' — CCTGCACCAT — GTTTTGAGGTTGAGTGAC — 3') and АТ1R-R (5' — AAAATAACAGGACA — AAAGGAGGCTAGGGAG — 3') (“LitTech”).
The program for amplification of the AT1R gene included initial denaturation at 93◦С for 60 seconds, 35 cycles (93◦ for 10 seconds, annealing specific for each pair of primers at 64◦С for 10 seconds, chain elongation at 72◦С for 20 seconds), and a final elongation at 72◦С for 60 seconds. Three types of amplification products were obtained: homozygote by allele 1, heterozygote, homozygote by allele 2 (allele 1 is the one indicated before the position of change, and allele 2 is the one indicated after the position of change).
Electrophoretic separation of amplicons was carried out by horizontal electrophoresis in cathode (-) to anode (+) direction in 3% agarose gel at 10-15 V per 1 cm of gel. The buffers used in electrophoresis included 1X Tris-acetate-EDTA (ТАЕ) solution, which was prepared from 50X ТАЕ buffer (0.04 М Tris-acetate, 0.002М ЕDТА, рН=8.3). The gels were stained with 1% ethidium bromide (EtBr) solution. The analyzed DNA fragments were observed as red bands when irradiated with UV light at wavelength of 310 nm.
To determine the plasma concentration of BNP in the study patients, a standard set was obtained from Peninsula Laboratories Inc. (USA). Blood sampling for analysis was done in fasting state at 8 a.m. from the cubital vein in the amount of 3 ml whole blood using a cooled syringe. The blood was collected into cooled polypropylene tubes containing EDTA (1 mg/1 ml of blood) and aprotinin (500 U per 1 mL of blood). Immediately after sampling, the whole blood was centrifuged for 15 minutes at 0°С (1600 rpm). The resulting plasma was transferred to eppendorf polypropylene tubes.
The maximum period for sampling the material preserved at 32oС before initiation of the enzyme immunoassay did not exceed 6 months. Twenty-five µL of primary antibodies to BNP were added to each well of the microplate, followed by incubation at +18-25oС for 60 min to ensure that the primary antibodies adhered to the well surfaces. Then, 50 µL of the standard solutions (with known BNP concentrations) and blood plasma samples were added with subsequent incubation at +18-250С for 120 min for binding with primary antibodies, followed by addition of 25 µL of biotinylated natriuretic peptide solution.
After incubation, the unbound BNP was washed out from the plate. The wells were washed from excessive unbound reagents, and then 100 µL of enzyme (streptavidin peroxidase) was added followed by incubation at +18-250С for 60 min for forming the AT-AG-AT-enzyme complex on a solid basis. The wells were washed again from the excessive unbound reagents, and 100 µL of substrate solution was added. The next incubation was done for 30 min at +18-25oС, and the reaction was finished by the addition of a stop-solution (100 µL). Photometric measurements were then captured at wavelength of 450 nm (differential filter 620 nm) on an automatic analyzer (“НumaReader Single”, Germany).
Enzyme-linked immunosorbent assay (ELISA) technique in determination of galectin-3 concentration
Plasma galectin-3 concentration of study patients was determined by ELISA using standard set of reagents produced by Bender MedSystems GmbH (Austria), and “Stat Fact 330” apparatus. The analysis was carried out in a clinical diagnostic laboratory of a private laboratory center, i.e. “Center B” (date of laboratory certification - March 2014). The laboratory director (S.O. Stepanets) was in charge. Detection of galectin-3 level was indicated in the attestation certificate of the laboratory; the reference value of plasma galectin-3 concentration was 0.0 - 2.28 ng/mL, with an average level of 0.54 ng/mL.
Blood sampling was done in fasting state from the cubital vein; blood was collected in the amount of 2 ml whole blood at 8 a.m. Then, 0.5 ml of plasma was separated, with the time between sampling of whole blood and plasma separation of less than 30 min. The separated plasma was frozen at -25oС. The wells of the microplate were washed twice with wash buffer and then 100 µL of sample solvent was added to the wells intended for the standards.
Standard dilutions on the microplate were prepared as follows: 100 µL of diluted standard of vascular endothelial (VE)-cadherin was added into the wells А1 and А2, and VE-cadherin standard was diluted by transferring 100 µL from well to well. After that, 100 µl from the last wells (G1 and G2) were removed, and 100 µL of the sample solvent was added into the well “Blank”. Then, 50 µL solvent for samples was added in the wells intended for samples and 50 µL of samples was added in those wells. The biotin conjugate was prepared and added into the wells “Blank”. After that, the microplate was covered with a plate seal and incubated for 2 hours at room temperature (18-25oС). Streptavidin conjugate was prepared. The well contents were completely removed and then washed with wash buffer for three times. Then, 100 µL of streptavidin conjugate was added into each well and the microplate was covered with a plate seal and incubated for 1 hour at room temperature (18-25oС). The well content was completely removed again and wells were washed with wash buffer for three times. Next, 100 µL of TMB substrate solution was added into each well, including the “Blank” well, and incubated for 10 minutes at room temperature. To stop the reaction, 100 µL of the Stop solution was added into each well, including the “Blank” well. The optical density of the wells at wavelength 450 nm and differentiated at filter 630 nm were then determined. Subsequently, the calibration curve was made on immune-enzyme analyzer, and the results and analysis were obtained.
The frequency distribution of the polymorphic genes in the population was tested according to the Hardy-Weinberg equilibrium law using GeneXpert calculator (http://gen-exp.ru/calculator_or.php) for calculation of a number of statistical parameters in case-control studies which use SNP Figure 1.
Evaluation of parameters of systemic and intracardiac hemodynamics was performed using echocardiography on echogram RADMIR ULTIMARA. Left ventricular muscle mass index (LVMMI) was calculated using Penn Convention formula. ECG registration was done according to conventional method using 12 standard leads. Blood pressure was measured according to recommendations of World Health Organization (WHO) experts.
Statistical methods
Statistical analyses included calculation of primary statistical indicators, detection of differences between groups by statistics, establishment of the relationship between variables using parametric (Pearson’s correlation) and nonparametric (Spearman’s correlation) correlation analysis, analysis of contingency tables, discriminate function analysis, and analysis of variance (ANOVA).
For quantitative indices, the primary statistical processing included calculation of the arithmetic mean (M), error of the mean (m), and mean root square deviation (σ). The differences between samples, distributed by the law of normal distribution, were estimated by Student t-test (t) for unrelated measurements.
The threshold levels of galectin-3 and BNP in blood plasma were calculated using the formula suggested by M.Yu. Antomonov (coauthored with V.M. Zhebel, O.O. Sakovych, G.V. Vilchynskiy, and O.O. SinghO [3.63]: Χ = [(M1 + 2 · m1) + (M2 — 2 · m2)] / 2, where Х is the threshold level of BNP M1 is the mean value of BNP or galectin-3 level in the group with no signs (relatively healthy), m1 is the error of M1, M2 is the mean value of BNP level in the group with signs (relatively sick), and m2 is the error of М2.
The following parameters were determined by the threshold level and discriminate function analysis: sensitivity (relative frequency of referring true-sick patient to the class of sick) and specificity (relative frequency of referring true-healthy patient to the class of healthy), infallibility (relative frequency of infallible decisions in regards to both true-sick and true-healthy individuals), false negative (relative frequency of referring true-sick patients to the class of healthy), and false positive (relative frequency of referring true-healthy patients to the class of sick).
The methods suggested by V.I. Yunkerov and S.G. Grigoriev 18 are the following: Sensitivity = (100 · а)/(а+b) (%), where а is the number of relatively sick individuals with signs, b is the number of relatively sick individuals with no signs; Specificity = (100 · d)/(c + d) (%), where с is the number of relatively healthy individuals with signs, and d is the number of relatively healthy individuals with no signs Infallibility = 100 · (а + d)/(а +b+ с+ d), (%) False negative reaction = (100 · b)/ (а+b), (%); and False positive reaction = (100 · с)/(c + d), (%).
Statistical analyses were done on a personal computer using the standard statistical package STATISTICA 10. Microsoft Excel was used for the primary preparation of tables and intermediate calculations.
Results
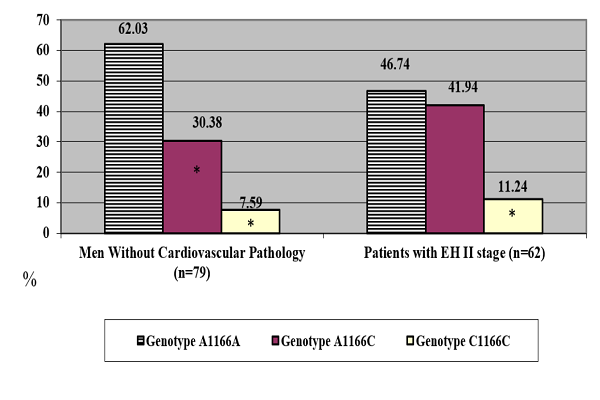
According the results obtained, the genotype frequency which includes the С allele of the AT1R gene was predominant in males with EH and LVH but no signs of CHF (Figure 2).
Because of the relatively small number of patients with the C1166C genotype, we combined the patients with A1166C and C1166C genotypes in the carriers of the C allele.
The frequency distribution of the genotypes of the AT1R gene was as follows: carriers of A1166A genotype- 46.77%, and carriers of the C allele- 53.23%. With regard to the analysis of the long-term course of EH in the subjects, the carriers of the various variants of the AT1R gene showed no dependence of its elongation on gene polymorphism (p>0.05).
One of the factors influencing the development of EH and BNP concentration is believed to be an increased body weight. The results obtained showed no difference in occurrence of males with normal weight, overweight and obesity when inheriting various genotypes of the AT1R gene (p>0.05).
A number of studies have demonstrated the potential use of blood BNP levels as a marker of LVH 19 and diastolic dysfunction 2021 in the carriers of AT1R polymorphic genes 22. However, no such data is available for galectin-3. Comparison within each genotype demonstrated the level of galectin-3 in blood plasma to be significantly higher in men with different stages of EH, as compared to those with no cardiovascular pathology (p <0.05) (Table 1).
However, males with EH, the carriers of the C allele, have significantly higher blood plasma concentration of galectin-3 than the carriers of the A1166A genotype (p<0.05).
| Groups | Blood plasma galectin-3 level | Blood plasma BNP level |
| Males with no cardiovascular athology ( =79) | 7.03 ± 0.12 ( =79) (1) | 21.74 ± 0.5 ( =79) (1) |
| Homozygotes of А1166А genotype | 6.82 ± 0.25 ( =49) (2) | 20.05 ± 2.02 ( =49) (2) |
| C allele carriers | 7.36 ± 0.40 ( =30) (3) | 21.88 ± 0.62 ( =30) (3) |
| Patients with stage II EH ( =62) | 21.31 ± 0.22 ( =62) (4) | 77.40 ± 2.85 ( =62) (4) |
| Homozygotes of А1166А genotype | 20.82 ± 0.51 ( =29) (5) | 56.56 ± 0.90 ( =29) (5) |
| C allele carriers | 22.87 ± 0.56 ( =33) (6) | 88.79 ± 2.97 ( =33) (6) |
| р | р3-2>0.05; р4-1<0.0001;р6-5<0.05 | р3-2>0.05; р4-1<0.0001;р5-2<0.0001; р6-3<0.0001; р6-5<0.05 |
The data obtained made it possible to calculate the threshold levels of galectin-3 for screening diagnosis of EH with myocardial hypertrophy in males (residents of Podillya region of Ukraine) to identify individuals who should undergo comprehensive examination of the heart, including ultrasound, and to investigate the causes of the steady increase of blood pressure:
— level of galectin-3 ≥ 21.01 ng/ml (sensitivity — 95.00%, specificity — 82.20%, infallibility — 89.23%, false-negative response — 5.00%, false-positive response — 11.82%) enables the diagnosis of stage II EH in the male population.
However, the probability of different concentrations of galectin-3 in blood plasma of the carriers of certain AT1R genotypes should be considered. At the same time, the threshold level of galectin-3 can differ in the carriers of polymorphic AT1R genotypes. The inheritance of the AA genotype of the AT1R gene proved to be associated with lower concentration of galectin-3 in blood plasma. Therefore, we decided to calculate the threshold levels for AA homozygotes of AT1R gene and C allele carriers (AC heterozygotes and CC homozygotes):
— level of galectin-3 ≥ 20.56 ng/ml (sensitivity — 76.77%, specificity — 87.97%, infallibility - 83.97%, false-negative response — 6.2%, false-positive response - 5.32%) enables the diagnosis of LVH in males, AA homozygotes of the AT1R gene;
— level of galectin-3 ≥ 24.89 ng/ml (sensitivity — 73.23%, specificity — 82.03%, infallibility — 78.16%, false-negative response — 3.80%, false-positive response — 4.67%) enables the diagnosis of EH with LVH in males, the carriers of the C allele of AT1R gene, and it is significantly higher than in AA homozygotes of AT1R gene.
Analysis of blood plasma galectin-3 level, in contrast to BNP, showed an opposite trend in men. In patients with stage II EH, an increased body weight was associated with higher plasma concentrations of galectin-3 (76.99 ± 5.07 ng/ml in subjects with BMI 25.0 - 29.9 kg/m2 (n = 21), vs. 5.79 ± 6.73 ng/ml in subjects with BMI ≥ 30.0 kg/m2 (n = 13)). The results obtained are consistent with data in the literature as well 2324.
Discussion
Large-scale genome-wide association studies (GWAS) have identified 107 gene loci that are associated with the development of cardiovascular diseases 25.
AT1R gene polymorphism can play an important role in heart remodeling since AT1R is responsible for the final pressor effects of the entire renin angiotensin system (RAAS) 26. A number of studies 27282213 have demonstrated the pathomorphological process in the form of hypertrophy to differ in the carriers of polymorphic variants of the AT1R gene. The authors established significantly higher carrier rate of C allele and C1166C genotype in patients with EH, and higher rate of A allele and A1166A genotype in the control group of patients. Taken together, the data strongly suggest that A allele and A1166A genotype play roles in reducing the risk of EH development. Indeed, the results obtained were consistent with data presented previously 276.
BNP is known to be the major factor, inhibiting the development of cardiac hypertrophy. Changes in its level can be considered as a pathognomonic sign in the process of cardiac remodeling 29. Moreover, galectin-3 has been demonstrated to be one of the active participants in the processes of collagen formation, inflammation, apoptosis, fibrosis and myocardial remodeling 22. The lowest plasma levels of BNP and galectin-3 in carriers of A1166A genotype of AT1R gene are indicative of a highly significant structural organization of AT1R gene in the functioning of RAAS. There is an association of this structural organization with plasma concentration of BNP in patients with EH.
EH patients (who are carriers of C allele genotypes) have significantly higher concentrations of BNP and galectin-3 in blood plasma than those who are homozygotes of A1166A genotype. This is indicative of a more pronounced RAAS activity in those with inherited A1166C and C1166C genotypes of the AT1R gene.
Thus, there is a probable connection between inheritance of A1166C and C1166C genotypes of AT1R gene and cardiovascular risk (i.e. presence of those genotypes could increase the risk for EH development. Moreover, the presence of those genotypes is probably associated not only with earlier development of EH, but also with rapid development of its associated complications.
It has been indicated in the literature 2730 that the level of BNP concentration in blood plasma is lower in obese patients than in subjects with normal body weight. Analysis of blood plasma galectin-3 level showed an opposite tendency; an increased body weight was associated with higher concentration of galectin-3.
Conclusions
In our study, the carriers of the C allele of the AT1R gene are predominantly men ranging from 40-60 years in age, residents of Podillya region of Ukraine, and ill with EH. The blood concentration of galectin-3 was significantly higher in EH patients with myocardial hypertrophy. Galectin-3 level was significantly higher in carriers of the C allele of AT1R gene. Indeed, its concentration is also higher in subjects with higher BMI. Thus, galectin-3 should be considered as a marker of myocardial hypertrophy.
Further studies of phenotypic markers are warranted to find the most simple, accessible and suitable biomarker in medical practice. As well, more up-to-date criteria for the risk of development of CHF associated with EH seem to be important and should be determined.
Open Access
This article is distributed under the terms of the Creative Commons Attribution License (CCBY4.0) which permits any use, distribution, and reproduction in any medium, provided the original author(s) and the source are credited.
List of abbreviations
ASH: American Society of Hypertension; AT1R: Polymorphism of angiotensin II type 1 receptor gene; BNP: Brain natriuretic peptide; BMI: Body mass index; BP: Bblood pressure; CHF: Chronic heart failure; DNA: Deoxyribonucleic acid; EDTA: Ethylenediaminetetraacetic acid; EH: Essential hypertension; ESC: European Society of Cardiology; EtBr: Ethidium bromide; GWAS: Genome-wide association studies; LVH: Left ventricular hypertrophy; LVMMI: Left ventricular muscle mass index; NUP: Natriuretic peptides; PCR: olymerase chain reaction; RAAS: Renin-angiotensin-aldosterone system; RNA: Ribonucleic acid; SNP: Single nucleotide polymorphism; TMB: 3,3',5,5'-Tetramethylbenzidine; WHO: World Health Organization; UV: Ultraviolet; USA: United States of America
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Competing interests
The authors declare no conflict of interest.
Funding
None
Authors' contributions
Phd Zhebel V.M conceptualized and designed the study, drafted the initial manuscript, and approved the final manuscript as submitted. Dr. Vita Ruzhanskaya carried out the initial analyses, reviewed and revised the manuscript, and approved the final manuscript as submitted. Dr. Viktor Sivak, Assistant Sakovych Helena, Assistant Yuliia Pashkova designed the data collection instruments, and coordinated and supervised data collection at two of the four sites, critically reviewed the manuscript, and approved the final manuscript as submitted. All authors approved the final manuscript as submitted and agree to be accountable for all aspects of the work.
Acknowledgments
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. We thank all patients for their participation in the investigation, and the doctors, nurses, and administrative staff of the Vinnytsia Regional Specialized Radiation Protection Dispensary and Military Medical Center of the Central Region of the Air Forces of Ukraine (Vinnytsia, Ukraine) that assisted with the study.
References
-
Ponikowski
P.,
Voors
A. A.,
Anker
S. D.,
Bueno
H.,
Cleland
J. G.,
Coats
A. J.,
2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: the Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. European Heart Journal.
2016;
37
:
2129-200
.
-
Yancy
C. W..
2017 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Journal of the American College of Cardiology.
2017;
1
:
1-195
.
-
Berezin
A.E..
Systolic and diastolic heart failure: two sides of the same process. Ukr. medicine chasopis.
2014;
3
:
91-96
.
-
Zhebel
VM.
On the genetic regulation of brain natriuretic peptide level in plasma of men with essential hypertension. Проблемы экологии и медицины.
2016;
20
.
-
Starzhynska
O.,
Zhebel
V.,
Blanar
O..
Polymorphism Angiotensin II type 1 receptor gene and endothelial dysfunction in patients with essential hypertension and congestive heart failure. The Canadian Journal of Cardiology.
2012;
28
:
115
.
-
Li
X.,
Zhang
P..
Genetic determinants of myocardial dysfunction. Journal of Medical Genetics.
2017;
54
:
1-10
.
-
Farrag
W.,
Eid
M.,
El-Shazly
S.,
Abdallah
M..
Angiotensin II type 1 receptor gene polymorphism and telomere shortening in essential hypertension. Molecular and Cellular Biochemistry.
2011;
351
:
13-8
.
-
Filippi
C. R. De.
Galectin-3 in heart failure – linking fibrosis, remodeling, and progression. US Cardiology.
2010;
7
:
67-70
.
-
Berezin
A. E..
Biomarkers in heart failure. Journal of Blood & Lymph.
2017;
7
:
1-8
.
-
de Boer
R. A.,
Edelmann
F.,
Cohen-Solal
A.,
Mamas
M. A.,
Maisel
A.,
Pieske
B..
Galectin-3 in heart failure with preserved ejection fraction. European Journal of Heart Failure.
2013;
15
:
1095-101
.
-
Berezin
A. E..
Galectin-3 as a phenotypic indicator of cardiovascular risk in patients with heart failure. Zaporizhia Medical Journal.
2013;
6
:
58-62
.
-
Adrian
L..
Is galectin-3 a promoter of ventricular dysfunction?. Revista Româna de Medicina de Laborator.
2018;
1
:
21-36
.
-
S. Hayet,
Khullar
Madhu.
Genetics and Pathophysiology of Essential HypertensionInTech Europe 2012.
Google Scholar -
Lip
G. Y.,
Felmeden
D. C.,
Li-Saw-Hee
F. L.,
Beevers
D. G..
Hypertensive heart disease. A complex syndrome or a hypertensive ‘cardiomyopathy’?. European Heart Journal.
2000;
21
:
1653-65
.
-
Maisch
B..
Ventricular remodeling. Cardiology.
1996;
87
:
2-10
.
-
Pashkova
I.,
Zhebel
V. M.,
Palahniuk
H. O.,
Sakovych
O. O.,
Starzhynska
O. L.,
Gumenyuk
A. F..
The BNP gene polymorphism as a regulator of brain natriuretic peptide plasma level in men with uncomplicated essential hypertension and left ventricular hypertrophy. Biological Markers and Guided Therapy.
2015;
2
:
13-23
.
-
Sakovich
O. O..
Inheritance of polymorphic genotypes of the angiotensin II receptor type II type and risk factors for the development of hypertonic disease in women living in the Vinnytsia region. Zaporozhye Medical Journal.
2011;
4
:
44-7
.
-
Yunkerov
V.I.,
S.G. Grigoriev.
St. Petersburg: Internal Medicine 2002.
Google Scholar -
Council
ESH Scientific,
Redon
Josep,
Narkiewicz
Krzysztof,
Nilsson
Peter M,
Burnier
Michel,
Viigimaa
Margus,
Ambrosioni
Ettore,
Coca
Antonio,
Olsen
Michael Hecht,
Schmieder
Roland E,
others
2013 ESH/ESC Guidelines for the management of arterial hypertension. European Heart Journal.
2013;
34
:
2159-2219
.
-
Ferreira
J. P.,
Duarte
K.,
Graves
T. L.,
Zile
M. R.,
Abraham
W. T.,
Weaver
F. A..
Natriuretic Peptides, 6-Min Walk Test, and Quality-of-Life Questionnaires as Clinically Meaningful Endpoints in HF Trials. Journal of the American College of Cardiology.
2016;
68
:
2690-707
.
-
Ledwidge
M.,
Gallagher
J.,
Conlon
C.,
Tallon
E.,
O’Connell
E.,
Dawkins
I..
Natriuretic peptide-based screening and collaborative care for heart failure: the STOP-HF randomized trial. Journal of the American Medical Association.
2013;
310
:
66-74
.
-
Madrigal-Matute
J.,
Lindholt
J. S.,
Fernandez-Garcia
C. E.,
Benito-Martin
A.,
Burillo
E.,
Zalba
G..
Galectin-3, a biomarker linking oxidative stress and inflammation with the clinical outcomes of patients with atherothrombosis. Journal of the American Heart Association.
2014;
3
:
566-74
.
-
Berezin
A. E..
Novel biomarkers in prediction of heart failure related outcomes: from bench to bedside. Internal Medicine (Tokyo, Japan).
2017;
7
:
236-7
.
-
Morrow David
A.,
Michelle
L..
Galectin-3 in Cardiovascular DiseaseA Possible Window Into Early Myocardial Fibrosis. Journal of the American College of Cardiology.
2012;
14
:
1257-8
.
-
Warren
Helen R,
Evangelou
Evangelos,
Cabrera
Claudia P,
Gao
He,
Ren
Meixia,
Mifsud
Borbala,
Ntalla
Ioanna,
Surendran
Praveen,
Liu
Chunyu,
Cook
James P,
others
Genome-wide association analysis identifies novel blood pressure loci and offers biological insights into cardiovascular risk. Nature genetics.
2017;
49
(3)
:
403
.
-
Bonnardeaux
A.,
Davies
E.,
Jeunemaitre
X.,
Féry
I.,
Charru
A.,
Clauser
E..
Angiotensin II type 1 receptor gene polymorphisms in human essential hypertension. Hypertension.
1994;
24
:
63-9
.
-
Butler
M. G..
Genetics of hypertension. Current status. Le Journal Medical Libanais.
2010;
58
:
175-8
.
-
Chandra
S.,
Narang
R.,
Sreenivas
V.,
Bhatia
J.,
Saluja
D.,
Srivastava
K..
Association of angiotensin II type 1 receptor (A1166C) gene polymorphism and its increased expression in essential hypertension: a case-control study. PLoS One.
2014;
9
:
e101502
.
-
Starzhinska
O. L..
Polymorphism of the renin-angiotensin-aldosterone system genes in cardiology. Biomedical and Biosocial Anthropology.
2013;
20
:
204-7
.
-
Fox
E. R.,
Musani
S. K.,
Singh
P.,
Bidulescu
A.,
Nagarajarao
H. S.,
Samdarshi
T. E..
Association of plasma B-type natriuretic peptide concentrations with longitudinal blood pressure tracking in African Americans: findings from the Jackson Heart Study. Hypertension.
2013;
61
:
48-54
.
Comments
Downloads
Article Details
Volume & Issue : Vol 5 No 8 (2018)
Page No.: 2633-2644
Published on: 2018-08-31
Citations
Copyrights & License

This work is licensed under a Creative Commons Attribution 4.0 International License.
Search Panel
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Google Scholar
Pubmed
Search for this article in:
Google Scholar
Researchgate
- HTML viewed - 3851 times
- Download PDF downloaded - 1274 times
- View Article downloaded - 0 times
 Biomedpress
Biomedpress




